Advertisements
Advertisements
प्रश्न
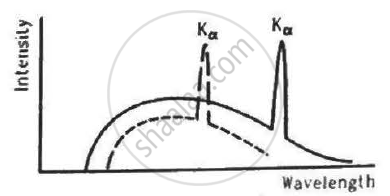
The figure shows the intensity-wavelength relations of X-rays coming from two different Coolidge tubes. The solid curve represents the relation for the tube A in which the potential difference between the target and the filament is VA and the atomic number of the target material is ZA. These quantities are VB and ZB for the other tube. Then,
विकल्प
VA > VB, ZA > ZB
VA > VB, ZA < ZB
VA < VB, ZA > ZB
VA > VB, ZA < ZB
उत्तर
VA > VB, ZA < ZB
It is clear from the figure that the X-ray of tube A has less cutoff wavelength than the X-ray of tube B.
`therefore lambda_A < lambda_B`
Using Moseley's Law,
`Z_A < Z_B`
`lambda ∝ 1/V` , where V is the voltage applied in the X-ray tube.
`therefore V_A > V_B`
APPEARS IN
संबंधित प्रश्न
Use the formula λm T= 0.29 cm K to obtain the characteristic temperature ranges for different parts of the electromagnetic spectrum. What do the numbers that you obtain tell you?
Name the high energetic invisible electromagnetic waves which help in the study of the structure of crystals
A wave has wavelength 50 Å.
- Name the wave.
- State its speed in vacuum.
- State its one use.
What do you understand by the statement, "Electromagnetic waves transport momentum"?
State Moseley's law
Which radiation is used for satellite communication?
What is the range of the wavelength of the following electromagnetic waves?
(a) Visible.
Give the range of wavelength of the electromagnetic waves visible to us.
State the approximate range of wavelength associated with visible light.
Name of physical quantity which remains same for microwaves of wavelength 1 mm and UV radiations of 1600 Å in vacuum.
Can Lα X-ray of one material have shorter wavelength than Kα X-ray of another?
An X-ray beam can be deflected
The X-ray beam emerging from an X-ray tube
For a given material, the energy and wavelength of characteristic X-rays satisfy
(a) E(Kα) > E(Kβ) > E(Kγ)
(b) E(Mα) > E(Lα) > E(Kα)
(c) λ(Kα) > λ(Kβ) > λ(Kγ)
(d) λ(Mα) > λ(Lα) > λ(Kα).
Find the energy, the frequency and the momentum of an X-ray photon of wavelength 0.10 nm.
(Use Planck constant h = 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
| Gamma rays | D | C | Visible light | B | A |
The above table shows different parts of the electromagnetic spectrum.
(a) Identify the parts of the spectrum marked as A, B, C and D.
(b) Which of the radiations A or B has the higher frequency?
(c) State two properties which are common to all parts of the electromagnetic spectrum.
(d) Name one source of each of the radiation of electromagnetic spectrum.
(e) Name one detector for each of the radiation.
(f) Name one use of each of the radiation.
State three properties of ultra-violet radiation similar to visible light.
Solve the numerical problem.
Calculate the frequency in MHz of a radio wave of wavelength 250 m. Remember that the speed of all EM waves in a vacuum is 3.0 × 108 m/s.
One requires 11eV of energy to dissociate a carbon monoxide molecule into carbon and oxygen atoms. The minimum frequency of the appropriate electromagnetic radiation to achieve the dissociation lies in ______.
In an atom X, electrons absorb the energy from an external source. This energy “excites” the electrons from a lower-energy level to a higher-energy level around the nucleus of the atom. When electrons return to the ground state, they emit photons.
The figure below is the energy level diagram of atom X with three energy levels, E1 = 0.00eV, E2 = 1.78eV and E3 = 2.95eV. The ground state is considered 0 eV for reference. The transition of electrons takes place between levels E1 and E2.
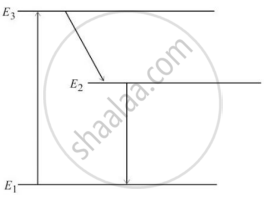
- What wavelength of radiation is needed to excite the atom to energy level E2 from E1?
- Suppose the external source has a power of 100 W. What would be the rate of photon emission?
