Advertisements
Advertisements
प्रश्न
Name or state the following:
An equation used in chemical calculations which gives a simultaneous effect of changes of temperature and pressure on the volume of a given mass of dry gas
उत्तर
Gas equation.
APPEARS IN
संबंधित प्रश्न
50 m3 of saturated vapour is cooled down from 30°C to 20°C. Find the mass of the water condensed. The absolute humidity of saturated water vapour is 30 g m−3 at 30°C and 16 g m−3 at 20°C.
What do you understand by gas?
What is diffusion? Give an example to illustrate it.
Fill in the blanks:
If the temperature is reduced to half, ………….. would also reduce to half.
Give reason for the following:
Volumes of gases are converted into s.t.p. conditions and then compared.
Estimate the average thermal energy of a helium atom at room temperature (27 °C).
Three vessels of equal capacity have gases at the same temperature and pressure. The first vessel contains neon (monatomic), the second contains chlorine (diatomic), and the third contains uranium hexafluoride (polyatomic).
Do the vessels contain an equal number of respective molecules?
Which of the following diagrams (Figure) depicts ideal gas behaviour?
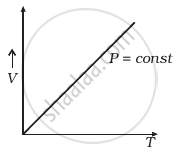 (a) |
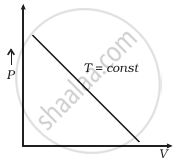 (b) |
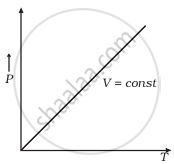 (c) |
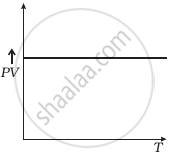 (d) |
Cooking gas containers are kept in a lorry moving with uniform speed. The temperature of the gas molecules inside will ______.
At room temperature, a diatomic gas is found to have an r.m.s. speed of 1930 ms-1. The gas is ______.
