Advertisements
Advertisements
प्रश्न
Name the type of isomerism shown by the following compounds:
[CU(NH3)4] [PtCl4] and [Pt(NH3)4] [CuCl4]
उत्तर
Coordination isomerism
APPEARS IN
संबंधित प्रश्न
What is the IUPAC name of 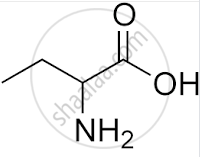
Write the IUPAC name of the following coordination compound:
[Co(NH3)6]Cl3
Write the IUPAC name of the following coordination compound:
[Pt(NH3)2Cl(NH2CH3)]Cl
Specify the oxidation number of the metal in the following coordination entity:
K3[Fe(CN)6]
Using IUPAC norms write the systematic name of the following:
[Pt(NH3)2Cl(NH2CH3)]Cl
Using IUPAC norms, write the systematic name of the following:
[Co(en)3]3+
Write down the IUPAC name of the following complex and indicate the oxidation state, electronic configuration and coordination number. Also, give the stereochemistry and magnetic moment of the complex:
[CrCl3(py)3]
Identify the compounds A, B, C, D, E, and F:
\[\ce{CH3COCH3->[Conc.HNO3][(O)]A}\]
\[\ce{->[SOCl2]B}\]
\[\ce{->[NH3]C}\]
\[\ce{->[LiAlH4]D}\]
\[\ce{->[HNO2]E}\]
\[\ce{->[CH3COCl]F}\]
Write the IUPAC name of the compound
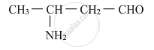
The formula Co(NH3)5CO3Cl could represent a carbonate or a chloride. Write the structures and names of possible isomers.
