Advertisements
Advertisements
प्रश्न
Specify the oxidation number of the metal in the following coordination entity:
K3[Fe(CN)6]
उत्तर
Let the oxidation number of metal be x.
Hence, 3 × (+1) + x + 6 × (−1) = 0
3 + x − 6 = 0
x − 3 = 0
∴ x = +3
APPEARS IN
संबंधित प्रश्न
Write the IUPAC name of [ Co(NO2)3(NH3)3 ].
Write the IUPAC names of the following coordination compounds: [Cr(NH3)3Cl3]
Write the IUPAC name of the given compound:
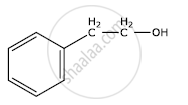
Specify the oxidation number of the metal in the following coordination entity:
[Co(H2O)(CN)(en)2]2+
Specify the oxidation number of the metal in the following coordination entity:
[PtCl4]2−
Specify the oxidation number of the metal in the following coordination entity:
[Cr(NH3)3Cl3]
Using IUPAC norms write the systematic name of the following:
[Pt(NH3)2Cl(NH2CH3)]Cl
Using IUPAC norms, write the systematic name of the following:
[Mn(H2O)6]2+
Using IUPAC norms, write the systematic name of the following:
[Ni(NH3)6]Cl2
Using IUPAC norms, write the systematic name of the following:
[Co(en)3]3+
Using IUPAC norms, write the systematic name of the following:
[Ni(CO)4]
Write down the IUPAC name of the following complex and indicate the oxidation state, electronic configuration and coordination number. Also, give the stereochemistry and magnetic moment of the complex:
[Co(NH3)5Cl]Cl2
Write down the IUPAC name of the following complex and indicate the oxidation state, electronic configuration and coordination number. Also, give the stereochemistry and magnetic moment of the complex:
[CrCl3(py)3]
Write down the IUPAC name of the following complex and indicate the oxidation state, electronic configuration and coordination number. Also, give the stereochemistry and magnetic moment of the complex:
K4[Mn(CN)6]
The oxidation number of cobalt in K[Co(CO)4] is
(i) +1
(ii) +3
(iii) −1
(iv) −3
Identify 'A' and 'B' and rewrite the reactions

How will you convert the following?
Aniline into N−phenylethanamide
Write the IUPAC name of the K3[Fe(C2O4)3].
The complex Hg[Co(CNS)4] is correctly named as ______.
