Advertisements
Advertisements
प्रश्न
Specify the oxidation number of the metal in the following coordination entity:
[Co(H2O)(CN)(en)2]2+
उत्तर
Let the oxidation number of the metal be x.
Hence, x + 0 + (−1) + (2 × 0) = +2
x = 2 + 1
∴ x = +3
APPEARS IN
संबंधित प्रश्न
What is the IUPAC name of 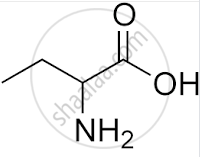
Write the IUPAC name of 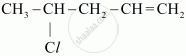
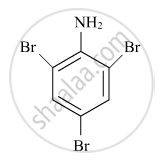
Write the IUPAC name of the given compound:
When a co-ordination compound CrCl3.6H2O is mixed with AgNO3, 2 moles of AgCl are precipitated per mole of the compound. Write
(i) Structural formula of the complex.
(ii) IUPAC name of the complex.
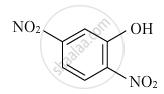
Write the IUPAC name of the given compound :
Write down the IUPAC name of the complex [Pt(en)2Cl2]2+. What type of isomerism is shown by this complex?
Write the IUPAC name of the following coordination compound:
[Co(NH3)6]Cl3
Specify the oxidation number of the metal in the following coordination entity:
[Cr(NH3)3Cl3]
Using IUPAC norms write the systematic name of the following:
[Co(NH3)6]Cl3
Using IUPAC norms, write the systematic name of the following:
[Ti(H2O)6]3+
Using IUPAC norms, write the systematic name of the following:
[Co(NH3)4Cl(NO2)]Cl
Using IUPAC norms, write the systematic name of the following:
[Ni(CO)4]
Write down the IUPAC name of the following complex and indicate the oxidation state, electronic configuration and coordination number. Also, give the stereochemistry and magnetic moment of the complex:
[Co(NH3)5Cl]Cl2
Write down the IUPAC name of the following complex and indicate the oxidation state, electronic configuration and coordination number. Also, give the stereochemistry and magnetic moment of the complex:
K4[Mn(CN)6]
Identify 'A' and 'B' and rewrite the reactions

Identify the compounds A, B, C, D, E, and F:
\[\ce{CH3COCH3->[Conc.HNO3][(O)]A}\]
\[\ce{->[SOCl2]B}\]
\[\ce{->[NH3]C}\]
\[\ce{->[LiAlH4]D}\]
\[\ce{->[HNO2]E}\]
\[\ce{->[CH3COCl]F}\]
Name the type of isomerism shown by the following compounds:
[CU(NH3)4] [PtCl4] and [Pt(NH3)4] [CuCl4]
The formula of the complex Iron (III) hexacyanidoferrate(II) is ______.
