Advertisements
Advertisements
प्रश्न
Name two electromagnetic waves of frequency smaller than that of violet light. State one use of each.
उत्तर
1) Ultraviolet radiations - 100 Å to 4000 Å
Use: For detecting purity of gems, eggs, ghee, etc.
2) X-rays - 0.1 Å to 100 Å
Use: For detecting fracture in bones, teeth, etc.
APPEARS IN
संबंधित प्रश्न
What do you understand by the statement, "Electromagnetic waves transport momentum"?
A wave has a wavelength of 10-3 nm. Name the wave.
The wavelength of X-rays is 0.01 Å. Calculate its frequency. State the assumption made, if any.
Frequencies of Kα X-rays of different materials are measured. Which one of the graphs in the figure may represent the relation between the frequency v and the atomic number Z ?

Find the energy, the frequency and the momentum of an X-ray photon of wavelength 0.10 nm.
(Use Planck constant h = 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
The Kβ X-ray of argon has a wavelength of 0.36 nm. The minimum energy needed to ionize an argon atom is 16 eV. Find the energy needed to knock out an electron from the K shell of an argon atom.
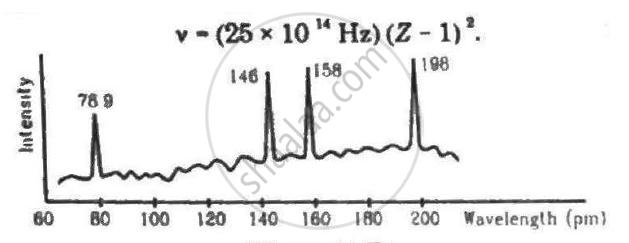
Continuous X-rays are made to strike a tissue paper soaked with polluted water. The incoming X-rays excite the atoms of the sample by knocking out the electrons from the inner shells. Characteristic X-rays are analysed and the intensity is plotted against the wavelength. Assuming that only Kα intensities are detected, list the elements present in the sample from the plot. Use Moseley's equation v − (25 × 1014Hz)(Z − 1)2.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Name the radiation of the electromagnetic spectrum which is used for the following:
Radar and Give the frequency range.
Answer briefly.
Can we produce a pure electric or magnetic wave in space? Why?
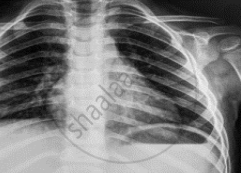
Name the electromagnetic radiation that has been used in obtaining the image below.