Advertisements
Advertisements
प्रश्न
Give the resonating structures of NO2 and N2O5.
उत्तर
NO2 :-
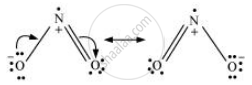
N2O5:-

APPEARS IN
संबंधित प्रश्न
Write balanced chemical equations for action of potassium permanganate on: Hydrogen
Why does NO2 dimerise?
What is the covalence of nitrogen in N2O5?
Bond angle in `PH_(4)^(+)` is higher than that in PH3. Why?
Write balanced chemical equations for action of potassium permanganate on Warm conc. sulphuric acid
Account for the following :
Bond angle in NH+4NH4+ is higher than NH3.
Which of the following statements is not correct for nitrogen?
Pure nitrogen is prepared in the laboratory by heating a mixture of:
Which of the following oxides of nitrogen is a coloured gas?
Which oxide of nitrogen is obtained on heating ammonium nitrate at 250°C?
On heating lead nitrate forms oxides of nitrogen and lead. The oxides formed are ______.
Name three oxoacids of nitrogen. Write the disproportionation reaction of that oxoacid of nitrogen in which nitrogen is in +3 oxidation state.
Assertion: \[\ce{HNO3}\] makes iron passive.
Reason: \[\ce{HNO3}\] forms a protective layer of ferric nitrate on the surface of iron.
On heating lead (II) nitrate gives a brown gas “A”. The gas “A” on cooling changes to colourless solid “B”. Solid “B” on heating with NO changes to a blue solid ‘C’. Identify ‘A’, ‘B’ and ‘C’ and also write reactions involved and draw the structures of ‘B’ and ‘C’.
Which oxide of nitrogen is obtained on heating ammonium nitrate at 250°C?
