Advertisements
Advertisements
प्रश्न
Saloni took a piece of burning charcoal and collected the gas evolved in a test tube.
(a) How will she find the nature of the gas?
(b) Write down word equations of all the reactions taking place in this process.
उत्तर
(a) Add a few drops of water in the test tube containing gas. Now, cover the test tube and shake it well. After shaking, test the solution with blue litmus and red litmus. It will turn blue litmus red. Thus, the gas is acidic in nature.
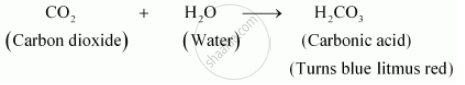
(b) Charcoal reacts with oxygen to form carbon dioxide gas.

Carbon dioxide reacts with water to form carbonic acid, which turns blue litmus paper red.
APPEARS IN
संबंधित प्रश्न
Mark ‘T’ if the statement is true and ‘F’ if it is false.
Generally, non-metals react with acids. ( )
Mention two uses of the following metals and non-metals
Iodine
Write ‘true’ or ‘false’ for the following statement.
Iodine acts as an antiseptic in the form of a tincture of iodine.
The electronic configuration of metal ‘A’ is 2,8,1 and that of metal ‘B’ is 2,8,2. Which of the two metals is more reactive? Write their reaction with dilute hydrochloric acid.
What is it made from? Why?
A pair of tongs.
Name the following:
A metal used in flexible pipes.
Soft metal : Na : : hard metal : _______
Halogen reacts with acid.
Assertion: Mercury is solid in room temperature.
Reason: Mercury is a non-metal.
X [2, 8, 7] and Y [2, 8, 2] are two elements. Using this information, complete the following:
Metal atoms tend to have a maximum of ______ electrons in the outermost shell.
