Advertisements
Advertisements
प्रश्न
Saloni took a piece of burning charcoal and collected the gas evolved in a test tube.
(a) How will she find the nature of the gas?
(b) Write down word equations of all the reactions taking place in this process.
उत्तर
(a) Add a few drops of water in the test tube containing gas. Now, cover the test tube and shake it well. After shaking, test the solution with blue litmus and red litmus. It will turn blue litmus red. Thus, the gas is acidic in nature.
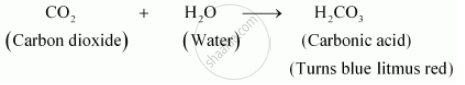
(b) Charcoal reacts with oxygen to form carbon dioxide gas.

Carbon dioxide reacts with water to form carbonic acid, which turns blue litmus paper red.
APPEARS IN
संबंधित प्रश्न
Mark ‘T’ if the statement is true and ‘F’ if it is false.
Sodium is a very reactive metal. ( )
Mark ‘T’ if the statement is true and ‘F’ if it is false.
Copper displaces zinc from zinc sulphate solution. ( )
Give a reason for Alkali metals are good reducing agents.
The atomic number of an element X is 12.
(a) What must an atom of X do to attain the nearest inert gas electron configuration?
(b) Which inert gas is nearest to X?
The atomic number of an element Y is 16.
(a) What must an atom of Y do to achieve the nearest inert gas electron arrangement?
(b) Which inert gas is nearest to Y?
Fill in the following blank with suitable word:
The electrical conductivity and melting point of an alloy is ........... than that of pure metals.
Fill in the blank spaces by choosing the correct words from the given list.
List: amalgam, chlorine, gallium, sodium chloride, silver chloride
______ is a liquid metal.
Give the general characteristics of:
Alkaline earth metals with reference to
(i) bonding
(ii) action of air
(iii) action of water
(iv) action of acid
Name the following:
A metal used in packaging.
Which metals are liquid at room temperature?
