Advertisements
Advertisements
प्रश्न
Show on a graph the variation of the de Broglie wavelength (λ) associated with an electron, with the square root of accelerating potential (V) ?
उत्तर
`lambda = 1.22/sqrtV`
`therefore lambda sqrtV=` const
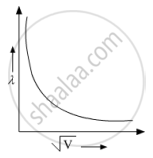
Hence, we get a hyperbola.
संबंधित प्रश्न
Calculate the de-Broglie wavelength of an electron moving with one-fifth of the speed of light.Neglect relativistic effects. (`h = 6.63 xx 10^(-34)` J.s, c= `3xx10^8`m/s, mass of electron = `9 xx 10^(-31) kg)`
State de Broglie hypothesis
A proton and an electron have same velocity. Which one has greater de-Broglie wavelength and why?
An electron of energy 150 eV has wavelength of 10-10m. The wavelength of a 0.60 keV electron is?
Light of wavelength 2000 Å falls on a metal surface of work function 4.2 eV.
What is the kinetic energy (in eV) of the fastest electrons emitted from the surface?
A particle is dropped from a height H. The de Broglie wavelength of the particle as a function of height is proportional to
Two bodies A and B having masses in the ratio of 3 : 1 possess the same kinetic energy. The ratio of linear momentum of B to A is:
For an electron accelerated from rest through a potential V, the de Broglie wavelength associated will be:
The de- Broglie wave length of an electron moving with a speed of 6.6 × 105 m/s is approximately
For production of characteristic of X-rays the electron trans mission is
