Advertisements
Advertisements
प्रश्न
Show on a graph the variation of the de Broglie wavelength (λ) associated with an electron, with the square root of accelerating potential (V) ?
उत्तर
`lambda = 1.22/sqrtV`
`therefore lambda sqrtV=` const
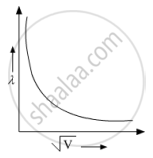
Hence, we get a hyperbola.
संबंधित प्रश्न
A proton and an alpha particle are accelerated through the same potential. Which one of the two has (i) greater value of de−Broglie wavelength associated with it and (ii) less kinetic energy. Give reasons to justify your answer.
A proton and an electron are accelerated by the same potential difference. Let λe and λpdenote the de Broglie wavelengths of the electron and the proton, respectively.
What is meant by diffraction of light?
Describe in brief what is observed when moving electrons are allowed to fall on a thin graphite film and the emergent beam falls on a fluorescent screen.
Answer the following question.
Obtain the expression for the ratio of the de-Broglie wavelengths associated with the electron orbiting in the second and third excited states of the hydrogen atom.
Light of wavelength 2000 Å falls on a metal surface of work function 4.2 eV.
What will be the change in the energy of the emitted electrons if the intensity of light with same wavelength is doubled?
Light of wavelength 2000 Å falls on a metal surface of work function 4.2 eV.
If the same light falls on another surface of work function 6.5 eV, what will be the energy of emitted electrons?
For production of characteristic of X-rays the electron trans mission is
The kinetic energy of electron in (electron volt) moving with the velocity of 4 × 106 m/s will be
An oil drop carrying a charge q has a mass m kg it is falling freely in air with terminal speed v. the electron field required to make the drop move upward with the same speed is
