Advertisements
Advertisements
प्रश्न
State whether the given chemical reaction is a redox reaction or not. Justify your answer.
\[\ce{MnO_2 + 4HCl->MnCl_2 +2H_2O + Cl_2}\]
उत्तर
The given reaction is an example of an oxidation-reduction reaction or redox reaction.
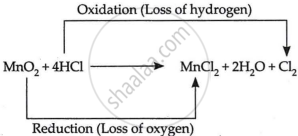
A substance is oxidised when it receives or loses hydrogen during a process. A material is decreased when it loses oxygen or absorbs hydrogen during a process. HCl loses hydrogen in the proposed process to generate Cl2. The provided reaction is thus a redox reaction.
संबंधित प्रश्न
Corrosion can be prevented by using ……………. solution.
The chemical formula for rust is.............. .
Fill in the following blanks with suitable words:
The addition of oxygen to a substance is called ......... whereas removal of oxygen is called ........
Fill in the following blanks with suitable words:
The addition of hydrogen to a substance is called ........ whereas removal of hydrogen is called .......
Give one example of an oxidation-reduction reaction which is also a combination reaction.
When copper powder is heated strongly in air, it forms copper oxide. Write a balanced chemical equation for this reaction. Name (i) substance oxidised, and (ii) substance reduced.
Hydrogen burns with a ______ flame and ______ sound is heard.
Explain the following in term of gain or loss of oxygen with two examples.
Oxidation
Name:
a liquid that is an oxidizing agent as well as a reducing agent.
The tarnishing of metals is due to the formation of ______.
