Advertisements
Advertisements
प्रश्न
Give the formula and describe the structure of a noble gas species which is isostructural with:
`BrO_3^(-)`
उत्तर १
XeO3 is isostructural to `BrO_3^(-)` and has a pyramidal molecular structure.
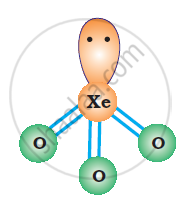
उत्तर २
In Br03– ion the central Br atom has 8 valence electrons (7 +1). Out of these, it shares 4 with two atoms of O forming Br = O bonds. Out of the remaining four .electrons, 2 are donated to the third O atom which accounts for its negative charge. The remaining 2 electrons constitute one lone pair. In order to minimise the force of repulsion, the structure of Br03– ion must be pyramidal. Br03– ion has (7 + 3 x 6 + 1) = 26 valence electrons and is isoelectronic as well as iso-structural with noble gas species Xe03 which has also 26(8 + 3 x 6) electrons.

APPEARS IN
संबंधित प्रश्न
Account for the following:
Helium is used in diving apparatus.
Draw the structures of the following molecules: XeF2
Why has it been difficult to study the chemistry of radon?
How are XeO3 and XeOF4 prepared?
Give the formula and describe the structure of a noble gas species which is isostructural with
`ICI_4^(-)`
Give the formula and describe the structure of a noble gas species which is isostructural with:
`IBr_2^(-)`
Answer the following.
List the uses of Neon and argon gases.
Complete the given chemical equations:
`2XeF_2 + 2H_2O->`
Draw the structures of the following
XeF6
Sulfur dioxide reacts with sodium hydroxide solution to form _______.
Explain the trend in the following atomic properties of group 16 elements:
Electron gain enthalpy
Which of the following fluorides does not exist?
Helium is preferred to be used in balloons instead of hydrogen because it is ____________.
Match the compounds given in Column I with the hybridisation and shape given in Column II and mark the correct option.
| Column I | Column II |
| (A) XeF6 | (1) sp3d3 – distorted octahedral |
| (B) XeO3 | (2) sp3d2 – square planar |
| (C) XeOF4 | (3) sp3 – pyramidal |
| (D) XeF4 | (4) sp3d2 – square pyramidal |
The order of increasing sizes of atomic radii among the elements O, S, Se and As is:
Noble gases are named because of their inertness towards reactivity. Identify an incorrect statement about them.
Match List - I with List - II:
| List - I | List - II | ||
| (Species) | (Number of lone pairs of electrons on the central atom) |
||
| (A) | XeF2 | (i) | 0 |
| (B) | XeO2F2 | (ii) | 1 |
| (C) | XeO3F2 | (iii) | 2 |
| (D) | XeF4 | (iv) | 3 |
Choose the most appropriate answer from the options given below:
Write two uses of neon.
