Advertisements
Advertisements
प्रश्न
Give the formula and describe the structure of a noble gas species which is isostructural with:
`BrO_3^(-)`
उत्तर १
XeO3 is isostructural to `BrO_3^(-)` and has a pyramidal molecular structure.
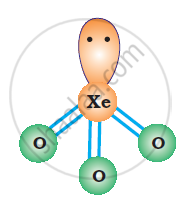
उत्तर २
In Br03– ion the central Br atom has 8 valence electrons (7 +1). Out of these, it shares 4 with two atoms of O forming Br = O bonds. Out of the remaining four .electrons, 2 are donated to the third O atom which accounts for its negative charge. The remaining 2 electrons constitute one lone pair. In order to minimise the force of repulsion, the structure of Br03– ion must be pyramidal. Br03– ion has (7 + 3 x 6 + 1) = 26 valence electrons and is isoelectronic as well as iso-structural with noble gas species Xe03 which has also 26(8 + 3 x 6) electrons.

APPEARS IN
संबंधित प्रश्न
Draw the structures of the following molecules: XeF4
Which noble gas is used in filling balloons for meteorological observations?
Complete the equation : XeF2+PF5 →
Complete the following equation:
XeF2 + H2O →
Why has it been difficult to study the chemistry of radon?
What inspired N. Bartlett for carrying out reaction between Xe and PtF6?
Which one of the following does not exist?
(i) XeOF4
(ii) NeF2
(iii) XeF2
(iv) XeF6
Complete the given chemical equations:
`2XeF_2 + 2H_2O->`
Complete the following reactions:
XeF6+2H2O ----->
Draw the structures of the following
XeF6
Write the electronic configuration of the following element:
Krypton (Z = 36)
Fill in the blanks by choosing the appropriate word/words from the brackets
(square pyramidal, electrical, 74; 26, sp3d2, sp3d, chemical, 68, 32, tetrahedral, yellow, white, iodoform, Lucas)
The geometry of XeOF4 molecule is ______ and the hybridisation of Xenon atom in the molecule is ________.
Sulfur dioxide reacts with sodium hydroxide solution to form _______.
Partial hydrolysis of XeF4 gives ____________.
In which of the following pairs, the two species are isostructural:
On partial hydrolysis, XeF6 gives ______.
\[\ce{XeF6 + H2O ->[Partial][Hydrolysis] \underline{}\underline{}\underline{}\underline{} + \underline{}\underline{}\underline{}\underline{}}\]
Discuss the trends in electronegativity and atomic radii for elements of group 16 17, 18.
