Advertisements
Advertisements
प्रश्न
The following question refers to one mole of chlorine gas.
If the relative atomic mass of chlorine is 35.5, what will be the mass of 1 mole of chlorine gas?
उत्तर
Molecular mass of chlorine (Cl2) = 71
∴ Mass of 1 mole of chlorine gas = 71 g
APPEARS IN
संबंधित प्रश्न
Give balanced chemical equations for the following conversions A, B, and C:
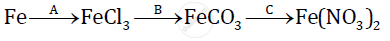
Calculate the percentage composition of oxygen in lead nitrate [Pb(NO3)2]. [Pb = 207, N= 14, O = 16]
Potassium nitrate on strong heating decomposes as under :
2KNO3 → 2KNO2 + O2
Calculate : Weight of potassium nitrite formed.
(K = 39, 0 = 16, N = 14)
Find the total percentage of oxygen in magnesium nitrate crystal Mg(NO3)2.6H2O.
[H = 1, N = 14, O = 16, Mg = 24]
What is the mass of nitrogen in 1000Kg of urea [CO(NH2)2] ?
[H = 1, C= 12, N= 14, O = 16]
Concentrated nitric acid oxidizes phosphorous to phosphoric acid according to the following equation :
P + 5HNO3 → H3PO4 + H2O + 5NO2
What mass of nitric acid will be consumed at the same time?
A flask contains 3.2g of sulphur dioxide. Calculate the following: The number of molecules of sulphur dioxide present in the flask.
Give two tests of the following:
Oxygen
Define or explain the term:
Vapour density
Calculate the mass of nitrogen supplied to soil by 5 kg of urea [CO(NH2)2].
[O = 16; N = 14; C = 12; H = 1]
