Advertisements
Advertisements
प्रश्न
The light emitted in the transition n = 3 to n = 2 in hydrogen is called Hα light. Find the maximum work function a metal can have so that Hα light can emit photoelectrons from it.
उत्तर
The Hα light can emit the photoelectrons if its energy is greater than or equal to the work function of the metal.
Energy possessed by Hα light (E) is given by
`E = 13.6 (1/n_1^2 - 1/n_2^2)eV`
`Here , n_1 = 2 , n_2 = 3`
`therefore E = 13.6 xx (1/4 - 1/4 )`
= `(13.6xx5)/36 = 1.89 eV`
= 1.90 eV
Hα light will be able to emit electron from the metal surface for the maximum work function of metal to be 1.90 eV.
APPEARS IN
संबंधित प्रश्न
An electron is orbiting in 5th Bohr orbit. Calculate ionisation energy for this atom, if the ground state energy is -13.6 eV.
(i) State Bohr's quantization condition for defining stationary orbits. How does the de Broglie hypothesis explain the stationary orbits?
(ii) Find the relation between three wavelengths λ1, λ2 and λ3 from the energy-level diagram shown below.
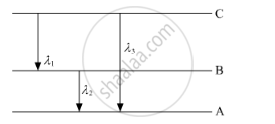
What is the maximum number of emission lines when the excited electron of an H atom in n = 6 drops to the ground state?
State Bohr's postulate to define stable orbits in the hydrogen atom. How does de Broglie's hypothesis explain the stability of these orbits?
Using Bohr’s postulates for hydrogen atom, show that the total energy (E) of the electron in the stationary states tan be expressed as the sum of kinetic energy (K) and potential energy (U), where K = −2U. Hence deduce the expression for the total energy in the nth energy level of hydrogen atom.
Evaluate Rydberg constant by putting the values of the fundamental constants in its expression.
Radiation coming from transition n = 2 to n = 1 of hydrogen atoms falls on helium ions in n = 1 and n = 2 states. What are the possible transitions of helium ions as they absorbs energy from the radiation?
What is the energy in joules released when an electron moves from n = 2 to n = 1 level in a hydrogen atom?
The energy associated with the first orbit of He+ is ____________ J.
According to Bohr's model of hydrogen atom, an electron can revolve round a proton indefinitely, if its path is ______.
Using Bohr's postulates derive the expression for the radius of nth orbit of the electron.
Consider two different hydrogen atoms. The electron in each atom is in an excited state. Is it possible for the electrons to have different energies but same orbital angular momentum according to the Bohr model? Justify your answer.
The ratio of the ionization energy of H and Be+3 is ______.
If a proton had a radius R and the charge was uniformly distributed, calculate using Bohr theory, the ground state energy of a H-atom when (i) R = 0.1 Å, and (ii) R = 10 Å.
The ground state energy of hydrogen atoms is -13.6 eV. The photon emitted during the transition of electron from n = 3 to n = 1 unknown work function. The photoelectrons are emitted from the material with a maximum kinetic energy of 9 eV. Calculate the threshold wavelength of the material used.
The wavelength in Å of the photon that is emitted when an electron in Bohr orbit with n = 2 returns to orbit with n = 1 in H atom is ______ Å. The ionisation potential of the ground state of the H-atom is 2.17 × 10−11 erg.
A hydrogen atom in its first excited state absorbs a photon of energy x × 10-2 eV and exited to a higher energy state where the potential energy of electron is -1.08 eV. The value of x is ______.
The energy of an electron in the first Bohr orbit of the H-atom is −13.6 eV. The energy value of an electron in the excited state of Li2+ is ______.
In hydrogen atom, transition from the state n = 6 to n = 1 results in ultraviolet radiation. Infrared radiation will be obtained in the transition ______.
The figure below is the Energy level diagram for the Hydrogen atom. Study the transitions shown and answer the following question:
- State the type of spectrum obtained.
- Name the series of spectrum obtained.

