Advertisements
Advertisements
प्रश्न
The radius of the nth orbit in the Bohr model of hydrogen is proportional to ______.
विकल्प
`n^2`
`1/n^2`
n
`1/n`
उत्तर
The radius of the nth orbit in the Bohr model of hydrogen is proportional to `underlinebb(n^2)`.
Explanation:
The radius of nth orbit is
`r_n = 0.53 xx n^2/ZÅ`
`r_n prop n^2`
APPEARS IN
संबंधित प्रश्न
(i) State Bohr's quantization condition for defining stationary orbits. How does the de Broglie hypothesis explain the stationary orbits?
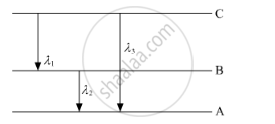
(ii) Find the relation between three wavelengths λ1, λ2 and λ3 from the energy-level diagram shown below.
If the velocity of the electron in Bohr’s first orbit is 2.19 × 106 ms-1, calculate the de Broglie wavelength associated with it.
A neutron moving with a speed υ strikes a hydrogen atom in ground state moving towards it with the same speed. Find the minimum speed of the neutron for which inelastic (completely or partially) collision may take place. The mass of neutron = mass of hydrogen = 1.67 × 10−27 kg.v
Suppose in an imaginary world the angular momentum is quantized to be even integral multiples of h/2π. What is the longest possible wavelength emitted by hydrogen atoms in visible range in such a world according to Bohr's model?
If the radius of first electron orbit in hydrogen atom be r then the radius of the fourth orbit ill be ______.
The energy of an electron in an excited hydrogen atom is - 3.4 eV. Calculate the angular momentum of the electron according to Bohr's theory. (h = 6.626 × 10-34 Js)
Hydrogen atom has only one electron, so mutual repulsion between electrons is absent. However, in multielectron atoms mutual repulsion between the electrons is significant. How does this affect the energy of an electron in the orbitals of the same principal quantum number in multielectron atoms?
A hydrogen atom in its first excited state absorbs a photon of energy x × 10-2 eV and exited to a higher energy state where the potential energy of electron is -1.08 eV. The value of x is ______.
A 20% efficient bulb emits light of wavelength 4000 Å. If the power of the bulb is 1 W, the number of photons emitted per second is ______.
[Take, h = 6.6 × 10-34 J-s]
How much is the angular momentum of an electron when it is orbiting in the second Bohr orbit of hydrogen atom?
