Advertisements
Advertisements
प्रश्न
The reagent which does not react with both, acetone and benzaldehyde.
विकल्प
Sodium hydrogensulphite
Phenyl hydrazine
Fehling’s solution
Grignard reagent
उत्तर
Fehling’s solution
Explanation:
Acetones belong to the ketonic functional category. To distinguish between aldehyde and ketone, utilise Fehling's solution. Carbonyl groups are found in sodium hydrogen sulphite, phenyl hydrazine and Grignard reagent. Ketones and aromatic aldehydes do not react with Fehling's solution.
APPEARS IN
संबंधित प्रश्न
On acid hydrolysis, propane nitrile gives.
How will you prepare the given compound from benzene? You may use any inorganic reagent and any organic reagent having not more than one carbon atom.
Phenylacetic acid
How will you bring about the following conversion in not more than two steps?
Benazaldehyde to α-Hydroxyphenylacetic acid
Name the reagents used in the following reactions:
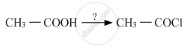
What is the action of following reagents on glucose?
1. Bromine water
2. Hydroxylamine
Which of the following substance produced acetaldehyde on dry distillation?
Match List - I with List - II.
| List - I | List - II | ||
| (a) |  \[\ce{->[CO,HCl][Anhyd. AlCl3/CuCl]}\] \[\ce{->[CO,HCl][Anhyd. AlCl3/CuCl]}\] |
(i) | Hell-Volhard-Zelinsky reaction |
| (b) |
\[\begin{array}{cc}
\ce{O}\phantom{.................}\\ ||\phantom{.................}\\ \ce{R - C - CH3 + NaOX ->} \end{array}\] |
(ii) | Gattermann-Koch reaction |
| (c) | \[\ce{R - CH2 - OH + R'COOH ->[Conc. H2SO4]}\] | (iii) | Haloform reaction |
| (d) | \[\ce{R - CH2COOH ->[(i) X2/Red P][(ii) H2O]}\] | (iv) | Esterification |
Choose the correct answer from the options given below.
The end product Y in the sequence of reaction:
\[\ce{RX ->[CN^-] X ->[NaOH] Y}\] is:
A compound 'X' with molecular formula C3H8O can be oxidised to a compound 'Y' with the molecular formula C3H6O2 'X' is most likely to be ______.
Fill in the blanks by choosing the appropriate words from those given in the brackets:
[stable, low, aldehyde, unstable, 6, 4, ethane, Clemmensen’s, 2, 3, carboxylic acid, high, propane, Rosenmund's]
The primary alcohols are easily oxidised first into ______ and then into ______.
