Advertisements
Advertisements
प्रश्न
How will you prepare the given compound from benzene? You may use any inorganic reagent and any organic reagent having not more than one carbon atom.
Phenylacetic acid
उत्तर

APPEARS IN
संबंधित प्रश्न
On acid hydrolysis, propane nitrile gives.
How is carbolic acid prepared from chlorobenzene?
Write the structures of A and B in the following reactions
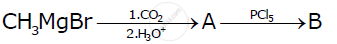
Show how the following compound can be converted to benzoic acid.
Acetophenone
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagent.
Tollens’ reagent
How will you prepare the given compound from benzene? You may use any inorganic reagent and any organic reagent having not more than one carbon atom.
m-Nitrobenzoic acid
How will you prepare the given compound from benzene? You may use any inorganic reagent and any organic reagent having not more than one carbon atom.
p-Nitrobenzoic acid
How will you bring about the following conversion in not more than two steps?
Benazaldehyde to α-Hydroxyphenylacetic acid
How is methoxy benzene prepared from carbolic acid?
Name the reagents used in the following reactions:
What is the action of following reagents on glucose?
1. Bromine water
2. Hydroxylamine
The functional group present in triacylglycerol is _______.
Match the reactions given in Column I with the suitable reagents given in Column II.
| Column I (Reactions) |
Column II (Reagents) |
| (i) Benzophenone Diphenylmethane | (a) \[\ce{LiAlH4}\] |
| (ii) Benzaldehyde 1-Phenylethanol | (b) \[\ce{DIBAL-H}\] |
| (iii) Cyclohexanone Cyclohexanol | (c) \[\ce{Zn(Hg)/Conc. HCl}\] |
| (iv) Phenyl benzoate Benzaldehyde | (d) \[\ce{CH3MgBr}\] |
Assertion: Aldehydes and ketones, both react with Tollen’s reagent to form silver mirror.
Reason: Both, aldehydes and ketones contain a carbonyl group.
Substitution of one alkyl group by replacing hydrogen of primary amines
The end product Y in the sequence of reaction:
\[\ce{RX ->[CN^-] X ->[NaOH] Y}\] is:
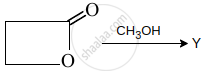
Y is:
