Advertisements
Advertisements
प्रश्न
The reason for using Aluminium in the alloy duralumin is ______.
विकल्प
Aluminium is brittle
Aluminium gives strength
Aluminium brings lightness
Aluminium lowers melting point
उत्तर
The reason for using Aluminium in the alloy duralumin is Aluminium brings lightness.
Explanation:
An alloy of aluminium, duralumin is used in making aircraft as it is light and corrosion-resistant.
APPEARS IN
संबंधित प्रश्न
Write the equation for the reaction that occurs at the cathode during the extraction of aluminum by electrolysis.
Give reason for the following:
Nitric acid can be stored in aluminium containers.
Aluminium is a more active metal than iron, but suffers less corrosion. Why?
Aluminium is extracted from its chief ore bauxite. The ore is first purified and then the metal is extracted from it by electrolytic reduction.
Write three balanced equations for the purification of bauxite by Hall's process.
Name the following :
A compound added to lower the fusion temperature of electrolytic bath in the extracton of aluminium.
Explain why :
In the electrolysis of alumina using the Hall Heroult's Process the electrolyte is covered with powdered coke.
During the concentration of bauxite ore, aluminium goes in ______ part because of its ______ nature.
Name the compound formed when: Bauxite reacts with sodium hydroxide.
State the relevant reason for the following:
A layer of powered coke is used over the electrolytic mixture in Hall Heroult’s process.
The following sketch illustrates the process of conversion of Alumina to Aluminium:
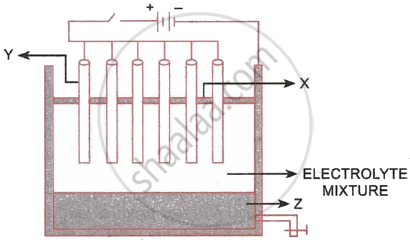
- Name the constituent of the electrolyte mixture which has a divalent metal in it.
- Name the powdered substances ‘X’ sprinkled on the surface of the electrolyte mixture.
- What is the name of the process?
- Write the reactions taking place at the electrodes ‘Y’ (anode) and ‘Z’ (cathode), respectively.
