Advertisements
Advertisements
प्रश्न
The work functions for potassium and caesium are 2.25 eV and 2.14 eV respectively. Is the photoelectric effect possible for either of them if the incident wavelength is 5180 Å?
[Given : Planck’s constant = 6.63 x 10–34 J.s.;
Velocity of light = 3 x 108 m/s; 1 eV = 1.6 x 10–19 J]
उत्तर
Given: (W0)P = 2.25 eV = 2.25 x 1.6 x 10-19 J = 3.6 x 10-19 J,
(W0)C = 2.14 eV = 2.14 x 1.6 x 10-19 J = 3.424 x 10-19 J,
λ = 5180 Å = 5.18 x 10-7 m
To find: Will the photoelectric effect occur for either of these elements with λ = 5180 Å
Formula: W0 = hv0
`(W_0)_p=h(v_0)_p`
`(v_0)_p=(W_0)_p/h=(3.6xx10^-19)/(6.63xx10^-34)`
`(v_0)_p=5.430xx10^14 Hz`
Similarly
`(v_0)_c=(W_0)_c/h=(3.424xx10^-19)/(6.63xx10^-34)`
`(v_0)_c=5.164 xx10^14Hz`
The corresponding frequency is given by,
`v_2=c/lambda=(3xx10^8)/(5.18xx10^-7)=3/5.18xx10^15`
`v_2=5.792xx10^14Hz`
for potassium
`5.792xx10^14Hz>5.430xx10^14Hz`
`i.e, v_2>(v_o)_p`
Photoelectric emission will take place when light of wavelength λ is incident on it. For caesium,
`5.792xx10^14Hz>5.16xx10^14Hz`
`i.e, v_2>(v_o)_c`
Photoelectric emission will take place when light of wavelength λ is incident on it. For λ= 5180Å wavelength, both potassium and caesium will exhibit photoelectric emission.
APPEARS IN
संबंधित प्रश्न
Draw a neat labelled circuit diagram of experimental arrangement for study of photoelectric effect.
Write three characteristic features in photoelectric effect that cannot be explained on the basis of wave theory of light, but can be explained only using Einstein's equation.
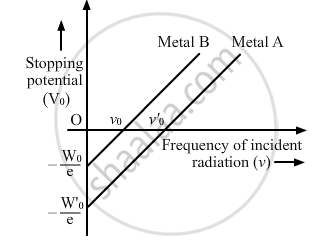
Sketch the graphs showing variation of stopping potential with frequency of incident radiations for two photosensitive materials A and B having threshold frequencies vA > vB.
(i) In which case is the stopping potential more and why?
(ii) Does the slope of the graph depend on the nature of the material used? Explain.
The photoelectric work function for a metal is 4.2 eV. If the stopping potential is 3V, find the threshold wavelength and maximum kinetic energy of emitted electrons.
(Velocity of light in air = 3 x 108m/s,
Planck's constant = 6·63 x10-34 J -s,
Charg.e ori electron = 1·6 x 10 -19 C)
Light of intensity ‘I’ and frequency ‘v’ is incident on a photosensitive surface and causes photoelectric emission. What will be the effect on anode current when (ii) the frequency of incident radiation is increased. In each case, all other factors remain the same. Explain, giving justification in each case.
Two monochromatic beams, one red and the other blue, have the same intensity. In which case (i) the number of photons per unit area per second is larger, (ii) the maximum kinetic energy of the photoelectrons is more? Justify your answer.
Draw a plot showing the variation of photoelectric current versus the intensity of incident radiation on a given photosensitive surface.
The graph shows the variation of stopping potential with frequency of incident radiation for two photosensitive metals A and B. Which one of the two has higher value of work-function? Justify your answer.
In an experiment of the photoelectric effect, the graph of maximum kinetic energy EK of the emitted photoelectrons versus the frequency v of the incident light is a straight line AB shown in Figure 6 below:
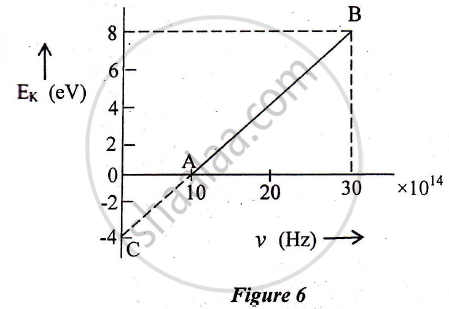
Find:
1) Threshold frequency of the metal
2) The work function of the metal.
3) Stopping potential for the photoelectrons emitted by the light of frequency `v = 30 xx 10^14 Hz`
Use Einstein's photoelectric equation to explain the observations from this graph ?
What change will you observe if intensity of incident radiation is changed but the frequency remains the same?
A beam of monochromatic radiation is incident on a photosensitive surface. Answer the following question giving reason :
On what factors does the number of emitted photoelectrons depend?
Draw a plot showing the variation of photoelectric current with collector plate potential for two different frequencies, v1 > v2, of incident radiation having the same intensity. In which case will the stopping potential be higher? Justify your answer.
Calculate the momentum of a photon of energy 6 x I 0-19 J.
Two metals A and B have work functions 4 eV and 6 eV respectively. Which metal has a lower threshold wavelength for photoelectric effect?
State how will you use this graph to detennine the value of Planck's constant.
If the frequency of the incident radiation is increased from 4 × 1015 Hz to 8 × 1015 Hz, by how much will the stopping potential for a given photosensitive surface go up?
Photoelectric effect is possible ______.
Light of wavelength 4000 Å is incident on two metals A and B. Which metal will emit photoelectrons, if their work functions are 3.8 e V and 1.6 e V respectively?
The phenomenon of photoelectric emission was observed by ______.
