Advertisements
Advertisements
प्रश्न
Using Bohr’s Theory of hydrogen atom, obtain an expression for the velocity of an electron in the nth orbit of an atom.
उत्तर
Let ‘e’, ‘m’, and ‘v’ be the charge, mass, and velocity of electrons, respectively, and ‘r’ is the radius of the orbit.
The positive charge on the nucleus is Ze, where Z is the atomic number (in case of hydrogen atom Z = 1).
As the electrostatic force of attraction provides the centripetal force.
`(mv^2)/r = 1/(4piepsilon_0) (("Ze") xx "e")/r^2`
`=> mv^2 = "Ze"^2/(4piepsilon_0r)`
`=> v = sqrt("Ze"^2/(4piepsilon_0" rm"))`
APPEARS IN
संबंधित प्रश्न
(i) State Bohr's quantization condition for defining stationary orbits. How does the de Broglie hypothesis explain the stationary orbits?
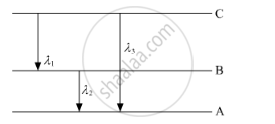
(ii) Find the relation between three wavelengths λ1, λ2 and λ3 from the energy-level diagram shown below.
The gravitational attraction between electron and proton in a hydrogen atom is weaker than the Coulomb attraction by a factor of about 10−40. An alternative way of looking at this fact is to estimate the radius of the first Bohr orbit of a hydrogen atom if the electron and proton were bound by gravitational attraction. You will find the answer interesting.
According to Maxwell's theory of electrodynamics, an electron going in a circle should emit radiation of frequency equal to its frequency of revolution. What should be the wavelength of the radiation emitted by a hydrogen atom in ground state if this rule is followed?
The light emitted in the transition n = 3 to n = 2 in hydrogen is called Hα light. Find the maximum work function a metal can have so that Hα light can emit photoelectrons from it.
Write postulates of Bohr’s Theory of hydrogen atom.
Which of the following is/are CORRECT according to Bohr's atomic theory?
(I) Energy is emitted when electron moves from a higher stationary state to a lower one.
(II) Orbits are arranged concentrically around the nucleus in an increasing order of energy.
(III) The energy of an electron in the orbit changes with time.
When an electric discharge is passed through hydrogen gas, the hydrogen molecules dissociate to produce excited hydrogen atoms. These excited atoms emit electromagnetic radiation of discrete frequencies which can be given by the general formula
`bar(v) = 109677 1/n_1^2 - 1/n_f^2`
What points of Bohr’s model of an atom can be used to arrive at this formula? Based on these points derive the above formula giving description of each step and each term.
Consider aiming a beam of free electrons towards free protons. When they scatter, an electron and a proton cannot combine to produce a H-atom ______.
- because of energy conservation.
- without simultaneously releasing energy in the from of radiation.
- because of momentum conservation.
- because of angular momentum conservation.
An electron in H-atom makes a transition from n = 3 to n = 1. The recoil momentum of the H-atom will be ______.
Hydrogen atom from excited state comes to the ground state by emitting a photon of wavelength λ. If R is the Rydberg constant then the principal quantum number n of the excited state is ______.
