Advertisements
Advertisements
प्रश्न
Using Sodium hydroxide solution, how would you distinguish: Ammonium sulphate from sodium sulphate.
उत्तर
Ammonium sulphate on reacting with sodium hydroxide liberates ammonia gas:
\[\ce{NH4)2SO4(aq) + 2NaOH(I) -> Na2SO4(aq) + 2NH3(I) + 2H2O(I)}\]
Sodium sulphate on reacting with sodium hydroxide undergoes the following change:
\[\ce{NaOH + Na2SO4 -> NaOH + Na2SO4}\]
However the products would be the same as the reactants, therefore providing no observation for a visible chemical reaction.
APPEARS IN
संबंधित प्रश्न
Identify the substance underlined, in the following case
Cation that does not form a precipitate with ammonium hydroxide but forms one with sodium hydroxide
What do you observe when caustic soda solution is added to the following solution, first a little and then in excess:
ZnSO4
Write a balanced equation for this reaction.
Using sodium hydroxide solution, how will you distinguish:
Iron (II) chloride from iron (III) chloride
Using Sodium hydroxide solution, how would you distinguish: Zinc nitrate solution from calcium nitrate solution.
Distinguish by adding:
Sodium hydroxide solution and ammonium hydroxide solution to calcium salt solution and lead salt solution.
Distinguish by adding:
Sodium hydroxide solution and ammonium hydroxide solution to copper salt solution and ferrous salt solution.
Distinguish by adding:
Sodium hydroxide solution and ammonium hydroxide solution to ferrous nitrate and lead nitrate.
What is observed when hot, concentrated caustic soda solution is added to zinc? Write a balanced equation.
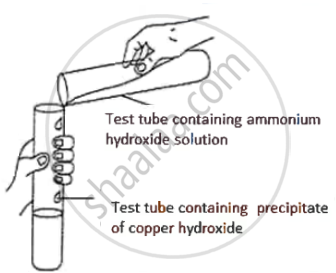
A student was asked to perform an experiment in the laboratory based on the instructions given:
Observe picture given below and state one observation for the experiment you would notice on mixing the given solution.
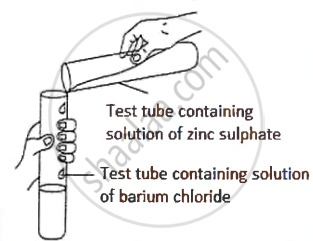
A student was asked to perform an experiment in the laboratory based on the instructions given:
Observe the picture given below and state one observation for the experiment you would notice on mixing the given solution.