Advertisements
Advertisements
प्रश्न
What are the important precautions?
उत्तर
Important precautions are -
(i)The lower end of thistle funnel must be dipped below the concentrated H2SO4.
(ii)Temperature should be maintained nearly 200°C because at higher temperature -
(a) The apparatus may break
(b) Fuel is wasted.
(c) Sodium sulphate formed in the reaction forms a crust on the surface which is difficult to remove.
APPEARS IN
संबंधित प्रश्न
Certain blank spaces are left in the following table and these are labelled as A, B, C, D
and E. Identify each of them
| Lab preparation of | Reactants used | Products formed | Drying Agent | Method of collection |
|
| 1 | HCl gas | NaCl + H2SO4 |  |
conc. H2SO4 | 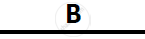 |
| 2 | NH3 gas |
|
Mg(OH)2 NH3 |
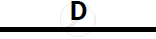 |
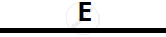 |
The following are pertaining to the laboratory preparation of hydrogen chloride gas.
Write the equation for its preparation mentioning the condition required.
Draw a labelled diagram for the laboratory preparation of hydrogen chloride gas and answer the following.
- Name the acid used. Why is this particular acid preferred to other acids?
- Give the balanced equation for the reaction.
- Name the drying agent used in drying hydrogen chloride gas.
- Phosphorous pentoxide and calcium oxide are good drying agents, but they cannot be used to dry hydrogen chloride gas. Why?
- Why is the direct absorption of \[\ce{HCl}\] gas in water not feasible?
- What arrangement is done to dissolve \[\ce{HCl}\] gas in the water?
Name the drying agents phosphorus pentoxide and calcium oxide are good drying agent but they cannot be used to dry hydrogen chloride gas. Why?
Explain why hydrogen chloride gas is not collected over water.
The given set up in the figure is for the preparation of an acid.
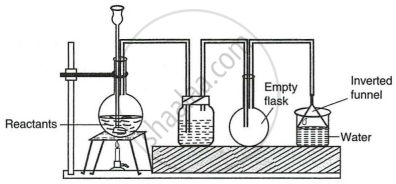
- Name the acid prepared by this method.
- Name the reactants used.
- Why an empty flask is used?
- What is the drying agent used? Why is this drying agent chosen?
- What is the role of the inverted funnel in the arrangement?
The drying agent used to dry \[\ce{HCl}\] gas is ______.
Draw a labelled diagram and explain the laboratory preparation of hydrogen chioride gas.
How will you prove that the gas prepared is HCI?
Explain, why the following statement is not correct:
Lead chloride can be prepared by adding dilute HCI to lead sulphate solution.
Write balanced equation for the reaction between dilute hydrochloric acid and sodium sulphite.
Write the equation for :
The preparation of hydrogen chloride from sodium chloride and sulphuric acid. State whether the sulphuric acid should be concentrated or dilute.
Give one test to distinguish between the following pair of chemicals.
Iron (III) Chloride solution and copper chloride solution.
Answer the following question related to the laboratory preparation of the hydrogen chloride gas:
Write the chemical equation.
In the laboratory preparation, HCl gas is dried by passing through ______.
State a relevant reason for the following:
Hydrogen chloride gas cannot be dried over quick lime.
