Advertisements
Advertisements
प्रश्न
What energy change would you Expect to take place in the molecules of a substance when it undergoes:
(i) a change in its temperature?
(ii) a change in itsstate without any change in its temperature?
उत्तर
(i) Inter molecular space changes.
(ii) Intermolecular space increases.
APPEARS IN
संबंधित प्रश्न
Name the law on which the principle of mixtures is based
Name the law on which principle of Calorimetry is based
By imparting heat to a body its temperature rises by 15°C. What is the corresponding rise in temperature on kelvin scale?
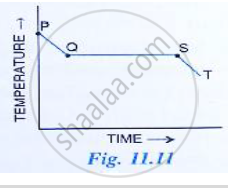
Fig 11. 11 shows the variation in temperature with time when some wax cools from the liquid phase to the solid phase.
(i) In which part of the curve, the wax is in liquid phase?
(ii) What does the part QS of the curve represent?
(iii) In which part of the curve, the wax will be the in the liquid as well as solid phase?
(iv) In which part of the curve, the wax is in solid phase?
Give scientific reasons for the following:
It is much easier to skate on rough ice than on glass.
Ice cream appears colder to the mouth than water at 0°C. Give a reason.
Fill in the following blank using suitable word:
The amount of heat required to change the state of a physical substance without any change of temperature is called .......... of the substance.
Find the final temperature when a mass of 80g of water at 100°C is mixed with a mass of 40g of water at 25°C.
