Advertisements
Advertisements
प्रश्न
What happens when Salicylic acid is treated with (CH3CO)2 O/H+?
उत्तर
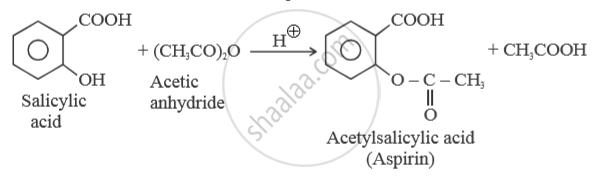
APPEARS IN
संबंधित प्रश्न
How is 1-nitropropane prepared from suitable oxime?
Although phenoxide ion has more number of resonating structures than carboxylate ion, carboxylic acid is a stronger acid than phenol. Give two reasons.
Which acid of the pair shown here would you expect to be stronger?
CH2FCH2CH2CO2H or CH3CHFCH2CO2H
Which acid of the pair shown here would you expect to be stronger?
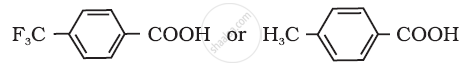
Write the reactions involved Hell-Volhard Zelinsky reaction?
Complete the following reaction sequence.
\[\begin{array}{cc}
\ce{O}\phantom{...............................................}\\
||\phantom{...............................................}\\
\ce{CH3 - C - CH3 ->[(i) CH3MgBr][H2O] (A) ->[Na metal][Ether] (B) ->[CH3 - Br] (C)}
\end{array}\]
Acidity of BF3 can be explained on the basis of which of the following concepts?
When propionamide reacts with Br2 in the presence of alkali the product is ______.
Na2CO3 cannot be used in place of (NH4)2CO3 for the precipitation of the V group because ______.
Formic acid and formaldehyde can be distinguished by treating with ______.
