Advertisements
Advertisements
Question
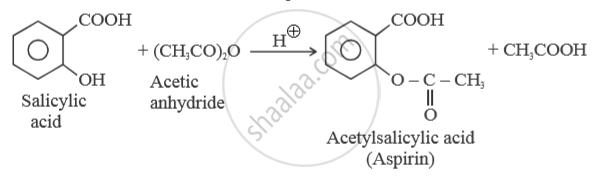
What happens when Salicylic acid is treated with (CH3CO)2 O/H+?
Solution
APPEARS IN
RELATED QUESTIONS
Although phenoxide ion has more number of resonating structures than carboxylate ion, carboxylic acid is a stronger acid than phenol. Give two reasons.
CH3CO2H or CH2FCO2H
Account for the following:
Carboxylic acid is a stronger acid than phenol.
Arrange the following compounds in increasing order of their property as indicated:
F - CH2COOH, O2N - CH2 COOH CH3 COOH,HCOOH - acid character.
Complete the following reaction sequence.
\[\begin{array}{cc}
\ce{O}\phantom{...............................................}\\
||\phantom{...............................................}\\
\ce{CH3 - C - CH3 ->[(i) CH3MgBr][H2O] (A) ->[Na metal][Ether] (B) ->[CH3 - Br] (C)}
\end{array}\]
Assertion: Formaldehyde is a planar molecule.
Reason: It contains sp2 hybridised carbon atom.
A mixture of benzaldehyde and formaldehyde on heating with 50% NaOH solution gives
When propionamide reacts with Br2 in the presence of alkali the product is ______.
Na2CO3 cannot be used in place of (NH4)2CO3 for the precipitation of the V group because ______.
Describe the action of alcoholic potassium hydroxide (alc. KOH) on isopropyl bromide.
