Advertisements
Advertisements
प्रश्न
What is meant by positive and negative deviations from Raoult's law and how is the sign of ΔmixH related to positive and negative deviations from Raoult's law?
उत्तर
According to Raoult’s law, the partial vapour pressure of each volatile component in any solution is directly proportional to its mole fraction. The solutions which obey Raoult’s law over the entire range of concentration are known as ideal solutions. If the solution does not obey Raoult’s law over the entire range of concentration, then it is called a non-ideal solution. The vapour pressure of such a solution is either higher or lower than that predicted by Raoult’s law. If the vapour pressure is higher, then the solution is said to exhibit a positive deviation, and if it is lower, then the solution is said to exhibit a negative deviation from Raoult’s law.
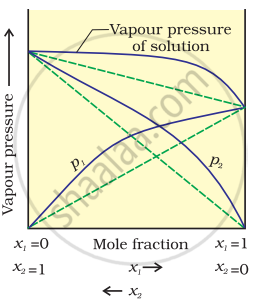
Vapour pressure of a two-component solution shows positive deviation from Raoult’s law
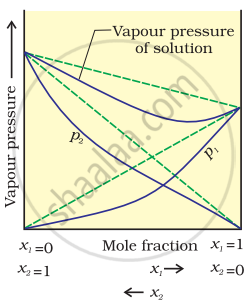
Vapour pressure of a two-component solution shows negative deviation from Raoult’s law
In the case of an ideal solution, the enthalpy of mixing the pure components to form the solution is zero.
ΔmixH = 0
In the case of solutions showing positive deviations, absorption of heat takes place.
∴ ΔmixH = Positive
In the case of solutions showing negative deviations, the evolution of heat takes place.
∴ ΔmixH = Negative
APPEARS IN
संबंधित प्रश्न
What role does the molecular interaction play in a solution of alcohol and water?
Heptane and octane form an ideal solution. At 373 K, the vapour pressures of the two liquid components are 105.2 kPa and 46.8 kPa respectively. What will be the vapour pressure of a mixture of 26.0 g of heptane and 35 g of octane?
What type of intermolecular attractive interaction exists in the pair of methanol and acetone?
The molality of a solution containing 1.8 g of glucose dissolved in 250 g of water is
State Raoult law and obtain the expression for lowering of vapour pressure when the nonvolatile solute is dissolved Insolvent.
The observed depression in the freezing point of water for a particular solution is 0.093°C calculate the concentration of the solution in molality. Given that molal depression constant for water is 1.86 K Kg mol-1.
For ideal solution the enthalpy of mixing of the pure components to form the solution is ____________.
For ideal solution the volume of mixing of the pure components to form the solution is ____________.
Which relationship is not correct?
Concentration terms such as mass percentage, ppm, mole fraction and molality are independent of temperature, however molarity is a function of temperature. Explain.
Why soda water bottle kept at room temperature fizzes on opening?
Upon increase in pressure for dissociation N2O4 into NO2, equilibrium shift towards
The pH of a solution of hydrochloric acid is 1.5. Calculate the concentration of the acid.
Lowering of vapour pressure of an aqueous solution of a non-volatile, non-electrolyte 1 molal aqueous solution at 100°C is ______.
If liquids A and B form an ideal solution then ______.
Assertion (A): The enthalpy of mixing Δmix H is equal to zero for an ideal solution.
Reason (R): For an ideal solution the interaction between solute and solvent molecules is stronger than the interactions between solute-solute or solvent-solvent molecules.
Suggest the most important type of intermolecular attractive interaction in the following pair.
I2 and CCl4
Suggest the most important type of intermolecular attractive interaction in the following pair.
NaClO4 and water
