Advertisements
Advertisements
प्रश्न
What is nucleus of an atom? Compare its size with that of the atom. Name its constitutents. How is the number of these constituents determined by the atomic number its atomic model.
उत्तर
The nucleus at the centre of atom, whose size is of the order of 10-15 m to 10-14 m.
The size of a nucleus is 10-5 to 10-4 times the size of an atom. It consists of protons and neutrons.
If Z is the atomic number and A is the mass number of an atom, then the atom contains Z number of electrons; Z number of protons and A - Z number of neutrons.
The atom is specified by the symbol
APPEARS IN
संबंधित प्रश्न
Write one use of cathode ray tube.
Name a gas caused by the Greenhouse effect
What is thermionic emission?
What do you mean by Atomic number
In the following fig. shows a radioactive source S in a thick lead container. The radiations pass through an electric field between the plates P and Q. Complete the diagram to show the paths of α , β and γ radiations.
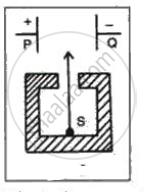
Why is the source S placed in a thick lead container?
State three safety precautions that you would take while handling the radioactive substances.
Why should a radioactive substance not be touched by hands?
An electrons emitter must have how much work function and melting point.
Unit of radioactivity is _______
In the following atoms, which one is a radioisotope? Give one use of this isotope.
O16, C14, N14, He4
