Advertisements
Advertisements
प्रश्न
What is nucleus of an atom? Compare its size with that of the atom. Name its constitutents. How is the number of these constituents determined by the atomic number its atomic model.
उत्तर
The nucleus at the centre of atom, whose size is of the order of 10-15 m to 10-14 m.
The size of a nucleus is 10-5 to 10-4 times the size of an atom. It consists of protons and neutrons.
If Z is the atomic number and A is the mass number of an atom, then the atom contains Z number of electrons; Z number of protons and A - Z number of neutrons.
The atom is specified by the symbol \[\ce{^A_Z X}\] where X is the chemical symbol for the element.
APPEARS IN
संबंधित प्रश्न
What are free electrons?
What do you mean by Mass number.
In following fig. shows a mixed source S of alpha and beta particles in a thick lead container. The particles pass through a magnetic field in a direction perpendicular to the plane of paper (inwards as shown by x). State and show in the diagram how the particles get affected.
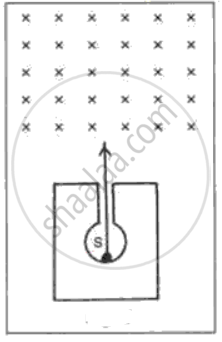
What are the types of emission?
How is a zinc sulphide screen affected when a cathode ray beam strikes it?
In a cathode ray tube state the purpose of covering cathode by thorium and carbon.
Are all the radiations viz. α, β and γ emitted in a single radioactive decay?
What is the value of the speed of gamma radiations in air or vacuum?
Mention four properties of Becquerel rays.
The half-life of a radioactive nuclide is 100 hours. The fraction of original activity that will remain after 150 hours would be ______.
