Advertisements
Advertisements
प्रश्न
What is the type of hybridisation of carbon atoms marked with star.
\[\ce{\overset{∗}{C}H3 - CH = CH - CH3}\]
उत्तर
The type of hybridization marked with the star in the given structure is:
\[\ce{CH3 - CH = CH - CH3}\]
The starred \[\ce{C}\] atom is sp3 hybridised.
APPEARS IN
संबंधित प्रश्न
What is the total number of sigma and pi bonds in the following molecules?
C2H2
What do you understand by bond pairs and lone pairs of electrons? Illustrate by giving one example of each type.
Isostructural species are those which have the same shape and hybridisation. Among the given species identify the isostructural pairs.
What is the type of hybridisation of carbon atoms marked with star.
\[\begin{array}{cc}
\phantom{.....}\ce{O}\\
\phantom{.....}||\\
\ce{\overset{∗}{C}H2 = CH - \overset{∗}{C} - O - H}
\end{array}\]
What is the type of hybridisation of carbon atoms marked with star.
\[\ce{CH3 - \overset{∗}{C}H2 - OH}\]
What is the type of hybridisation of carbon atoms marked with star.
\[\begin{array}{cc}
\phantom{..........}\ce{O}\\
\phantom{..........}||\\
\ce{CH3 - CH2 - \overset{∗}{C} - H}
\end{array}\]
What is the type of hybridisation of carbon atoms marked with star.
\[\ce{CH3 - \overset{∗}{C} ≡ CH}\]
BF3 is a planar and electron-deficient compound. Hybridization and the number of electrons around the central atom, respectively are ______.
In the given reaction,
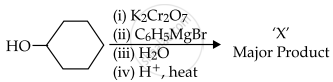
the number of sp2 hybridised carbon (s) in compound 'X' is ______.
The hybridisation of carbanion is:
