Advertisements
Advertisements
प्रश्न
What do you understand by bond pairs and lone pairs of electrons? Illustrate by giving one example of each type.
उत्तर १
When two atoms combine by sharing their one or more valence electrons, a covalent bond is formed between them.
The shared pairs of electrons present between the bonded atoms are called bond pairs. All valence electrons may not participate in bonding. The electron pairs that do not participate in bonding are called lone pairs of electrons.
For example, in C2H6 (ethane), there are seven bond pairs but no lone pair present.
\[\begin{array}{cc}
\ce{H}\phantom{....}\ce{H}\\
|\phantom{.....}|\\
\ce{H - C - C - H}\\
|\phantom{.....}|\\\ce{H}\phantom{....}\ce{H}
\end{array}\]
In H2O, there are two bond pairs and two lone pairs on the central atom (oxygen).
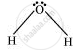
उत्तर २
The electron pair involved in sharing between two atoms during covalent bonding is called shared pair or bond pair. At the same time, the electron pair which is not involved in sharing is called lone pair of electrons.
For example, In `"CH"_4` 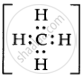 there are only 4 bond pairs, but in `"H"_2"O"`,
there are only 4 bond pairs, but in `"H"_2"O"`, 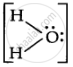 there are two bond pairs and two lone pairs.
there are two bond pairs and two lone pairs.
APPEARS IN
संबंधित प्रश्न
Apart from tetrahedral geometry, another possible geometry for CH4 is square planar with the four H atoms at the corners of the square and the C atom at its centre. Explain why CH4 is not square planar?
Draw a diagram showing the formation of a double bond and a triple bond between carbon atoms in C2H4 and C2H2 molecules.
What is the total number of sigma and pi bonds in the following molecules?
C2H2
Distinguish between a sigma and a pi bond.
What is the total number of sigma and pi bonds in the following molecules?
C2H4
Which of the following angle corresponds to sp2 hybridisation?
Predict the shapes of the following molecules on the basis of hybridisation.
\[\ce{BCl3, CH4 , CO2, NH3}\]
Match the species in Column I with the type of hybrid orbitals in Column II.
| Column I | Column II |
| (i) \[\ce{SF4}\] | (a) sp3d2 |
| (ii) \[\ce{IF5}\] | (b) d2sp3 |
| (iii) \[\ce{NO^{+}2}\], | (c) sp3d |
| (iv) \[\ce{NH^{+}4}\], | (d) sp3 |
| (e) sp |
Match the shape of molecules in Column I with the type of hybridisation in Column II.
| Column I | Column II |
| (i) Tetrahedral | (a) sp2 |
| (ii) Trigonal | (b) sp |
| (iii) Linear | (c) sp3 |
What is the type of hybridisation of carbon atoms marked with star.
\[\begin{array}{cc}
\phantom{.....}\ce{O}\\
\phantom{.....}||\\
\ce{\overset{∗}{C}H2 = CH - \overset{∗}{C} - O - H}
\end{array}\]
What is the type of hybridisation of carbon atoms marked with star.
\[\ce{\overset{∗}{C}H3 - CH = CH - CH3}\]
What is the type of hybridisation of carbon atoms marked with star.
\[\ce{CH3 - \overset{∗}{C} ≡ CH}\]
BF3 is a planar and electron-deficient compound. Hybridization and the number of electrons around the central atom, respectively are ______.
In which of the following species S atom assumes sp3 hybrid state?
(I) (SO3)
(II) SO2
(III) H2S
(IV) S8
