Advertisements
Advertisements
प्रश्न
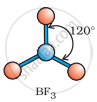
Which of the following angle corresponds to sp2 hybridisation?
विकल्प
90°
120°
180°
109°
उत्तर
120°
Explanation:
For a molecule having a hybridization of sp2, its geometry in plane is trigonal planar. For a molecule having a trigonal planar geometry, its bond angle will be 120°
APPEARS IN
संबंधित प्रश्न
Apart from tetrahedral geometry, another possible geometry for CH4 is square planar with the four H atoms at the corners of the square and the C atom at its centre. Explain why CH4 is not square planar?
What is the total number of sigma and pi bonds in the following molecules?
C2H2
Distinguish between a sigma and a pi bond.
The types of hybrid orbitals of nitrogen in \[\ce{NO^{+}2}\] , \[\ce{NO^{-}3}\] and \[\ce{NH^{+}4}\] respectively are expected to be ______.
Predict the shapes of the following molecules on the basis of hybridisation.
\[\ce{BCl3, CH4 , CO2, NH3}\]
Match the species in Column I with the type of hybrid orbitals in Column II.
| Column I | Column II |
| (i) \[\ce{SF4}\] | (a) sp3d2 |
| (ii) \[\ce{IF5}\] | (b) d2sp3 |
| (iii) \[\ce{NO^{+}2}\], | (c) sp3d |
| (iv) \[\ce{NH^{+}4}\], | (d) sp3 |
| (e) sp |
What is the type of hybridisation of carbon atoms marked with star.
\[\ce{CH3 - \overset{∗}{C}H2 - OH}\]
What is the type of hybridisation of carbon atoms marked with star.
\[\begin{array}{cc}
\phantom{..........}\ce{O}\\
\phantom{..........}||\\
\ce{CH3 - CH2 - \overset{∗}{C} - H}
\end{array}\]
What is the type of hybridisation of carbon atoms marked with star.
\[\ce{\overset{∗}{C}H3 - CH = CH - CH3}\]
BF3 is a planar and electron-deficient compound. Hybridization and the number of electrons around the central atom, respectively are ______.
