Advertisements
Advertisements
प्रश्न
Match the species in Column I with the type of hybrid orbitals in Column II.
| Column I | Column II |
| (i) \[\ce{SF4}\] | (a) sp3d2 |
| (ii) \[\ce{IF5}\] | (b) d2sp3 |
| (iii) \[\ce{NO^{+}2}\], | (c) sp3d |
| (iv) \[\ce{NH^{+}4}\], | (d) sp3 |
| (e) sp |
उत्तर
| Column I | Column II |
| (i) \[\ce{SF4}\] | (c) sp3d |
| (ii) \[\ce{IF5}\] | (a) sp3d2 |
| (iii) \[\ce{NO^{+}2}\], | (e) sp |
| (iv) \[\ce{NH^{+}4}\], | (d) sp3 |
Explanation:
(i) \[\ce{SF4}\] has a total of 34 valence electrons. Sulphur has 6 valence electrons and each fluoride has 7 valence electrons. The central atom sulphur will thus share one electron with all the four fluoride ions and thus make four bonds. A lone pair of electrons (two electrons) is left on the sulphur atom. Thus, the sulphur atom is surrounded by five electron pairs, four bond pairs and one lone pair. Having five electron pairs surrounding sulphur, it will have a trigonal bipyramidal geometry with sp3d hybridization. Due to the lone pair of electrons, bond pair-lone pair repulsion occurs which causes the bond angle to change. This change in the bond angle causes the distortion in the geometry and changes it into see-saw shape.
(ii) \[\ce{IF5}\] has the total of 42 valence shell electrons, 7 valence electrons in iodine and 7 in each of the fluoride ions. The central iodine atom will thus make five sigma bonds with chlorine and a lone pair of electrons will be left on the iodine atom. The six electron pairs thus will have a hybridization of sp3d2 and octahedral geometry. Due to the presence of lone pairs of electrons on the iodine, lone pair-bond pair repulsion occurs and the structure forms a distorted octahedral geometry or a square pyramidal geometry.
(iii) In \[\ce{NO^{+}2}\], nitrogen forms two sigma bonds and two pi-bonds with the oxygen atom. The oxygen atoms are arranged in an angle of 180° with the nitrogen atoms. The s-orbital of the nitrogen forms a hybrid with the p-orbital of the nitrogen atom, thereby forming two sp hybridized hybrid orbitals, having a linear geometry.
(iv) \[\ce{NH^{+}4}\] has the total of 8 valence electrons. Due to the presence of positive charge, nitrogen has 4 valence electrons and hydrogen has one in every four hydrogen atoms. The four valence electrons in nitrogen make sigma bonds with the four hydrogen atoms, thereby making a tetrahedral geometry and having sp3 hybridized orbitals.
APPEARS IN
संबंधित प्रश्न
Apart from tetrahedral geometry, another possible geometry for CH4 is square planar with the four H atoms at the corners of the square and the C atom at its centre. Explain why CH4 is not square planar?
Draw a diagram showing the formation of a double bond and a triple bond between carbon atoms in C2H4 and C2H2 molecules.
What is the total number of sigma and pi bonds in the following molecules?
C2H2
What do you understand by bond pairs and lone pairs of electrons? Illustrate by giving one example of each type.
Predict the shapes of the following molecules on the basis of hybridisation.
\[\ce{BCl3, CH4 , CO2, NH3}\]
Match the shape of molecules in Column I with the type of hybridisation in Column II.
| Column I | Column II |
| (i) Tetrahedral | (a) sp2 |
| (ii) Trigonal | (b) sp |
| (iii) Linear | (c) sp3 |
Discuss the concept of hybridisation. What are its different types in a carbon atom.
What is the type of hybridisation of carbon atoms marked with star.
\[\ce{CH3 - \overset{∗}{C} ≡ CH}\]
In the given reaction,
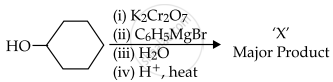
the number of sp2 hybridised carbon (s) in compound 'X' is ______.
The hybridisation of carbanion is:
