Advertisements
Advertisements
प्रश्न
Match the shape of molecules in Column I with the type of hybridisation in Column II.
| Column I | Column II |
| (i) Tetrahedral | (a) sp2 |
| (ii) Trigonal | (b) sp |
| (iii) Linear | (c) sp3 |
उत्तर
| Column I | Column II |
| (i) Tetrahedral | (c) sp3 |
| (ii) Trigonal | (a) sp2 |
| (iii) Linear | (b) sp |
Explanation:
(i) A tetrahedral molecule has four electron pairs and these make sigma bonds with each other. The s and p orbitals overlap with each other thus forming a sp3 hybridized molecule.
(ii) There are two possibilities for the central atom to have sp2 hybridization. Either all the bonds are in place i.e, the bond pairs are all sigma bonds or pi bonds or there are only two bonds and one lone pair of electrons.
(iii) The sp hybridization involves the mixing of the valence electrons in s orbital with another valence electron in the p orbital which yields two sp orbitals which are oriented in a linear geometry.
APPEARS IN
संबंधित प्रश्न
What do you understand by bond pairs and lone pairs of electrons? Illustrate by giving one example of each type.
Isostructural species are those which have the same shape and hybridisation. Among the given species identify the isostructural pairs.
Which of the following angle corresponds to sp2 hybridisation?
What is the type of hybridisation of carbon atoms marked with star.
\[\ce{CH3 - \overset{∗}{C}H2 - OH}\]
What is the type of hybridisation of carbon atoms marked with star.
\[\begin{array}{cc}
\phantom{..........}\ce{O}\\
\phantom{..........}||\\
\ce{CH3 - CH2 - \overset{∗}{C} - H}
\end{array}\]
What is the type of hybridisation of carbon atoms marked with star.
\[\ce{CH3 - \overset{∗}{C} ≡ CH}\]
BF3 is a planar and electron-deficient compound. Hybridization and the number of electrons around the central atom, respectively are ______.
In the given reaction,
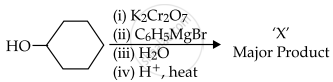
the number of sp2 hybridised carbon (s) in compound 'X' is ______.
In which of the following species S atom assumes sp3 hybrid state?
(I) (SO3)
(II) SO2
(III) H2S
(IV) S8
The hybridisation of carbanion is:
