Advertisements
Advertisements
प्रश्न
What is meant by atomicity ? Explain with two examples.
उत्तर
Atomicity refers to the total number of atoms present in one molecule of an element.
For example, argon is a noble gas and exists in a free state. One molecule of argon comprises only one atom. Therefore, the atomicity of argon is one.
Similarly, a molecule of chlorine gas has two atoms and it exists as a Cl2 molecule. Therefore, the atomicity of chlorine is two.
APPEARS IN
संबंधित प्रश्न
Write down the name of a compound represented by the following formula:
KNO3
Give the name of the element present in the following compound:
Hydrogen bromide
State the term.
The number of atoms present in a molecule of an element _________
Define: Atomic weight :
In what form do noble gases occur in nature?
Calculate the mass of 3.011 × 1024 atoms of carbon.
Explain the term – molecules. Give three examples of atoms of the same element forming a molecule. State the atomicity of the same.
Give a reason why –
Helium is considered monoatomic, but hydrogen a diatomic molecule.
Classify the following molecules as the molecules of element or compound
| 1. |  |
|
| 2. |  |
|
| 3. | 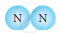 |
|
| 4. | 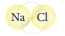 |
Write any two classifications of a molecule.
