Advertisements
Advertisements
Question
What is meant by atomicity ? Explain with two examples.
Solution
Atomicity refers to the total number of atoms present in one molecule of an element.
For example, argon is a noble gas and exists in a free state. One molecule of argon comprises only one atom. Therefore, the atomicity of argon is one.
Similarly, a molecule of chlorine gas has two atoms and it exists as a Cl2 molecule. Therefore, the atomicity of chlorine is two.
APPEARS IN
RELATED QUESTIONS
Give the name of the element present in the following compound:
Potassium sulphate
What is the atomicity of the following ?
- Oxygen
- Ozone
- Neon
- Sulphur
- Phosphorus
- Sodium
Why are the symbols and formulae of substances important?
Give a reason why –
Ozone is considered triatomic, but phosphorus a tetratomic molecule.
State the atomicity of the following:
Oxygen
Classify the following molecules as the molecules of element or compound
| 1. |  |
|
| 2. |  |
|
| 3. | 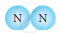 |
|
| 4. | 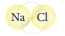 |
Which of the following is a triatomic molecule?
If a molecule is made of similar kinds of atoms, then it is called ______ atomic molecule.
Two elements sometimes can form more than one compound.
Write any two classifications of a molecule.
