Advertisements
Advertisements
प्रश्न
When an ammonium salt is warmed with a sodium hydroxide solution, a gas is evolved. State three ways in which you can identify this gas.
उत्तर
Three ways in which ammonia gas can be identified is:
It has a sharp characteristic odour When a glass rod dipped in HCl is brought in contact with the gas white colour fumes of ammonium chloride are formed It turns moist red litmus blue, moist turmeric paper brown and phenolphthalein solution pink.
APPEARS IN
संबंधित प्रश्न
Is ammonia more dense or less dense than air?
Complete the following equation. What property of ammonia is illustrated by the reaction given below.
2NH3 + 3CuO →
The diagram shows a simple arrangement of the fountain experiment:
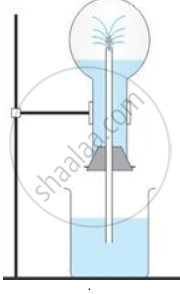
(i) Name the two gases you have studied which can be used in this experiment
(ii) What is the common properly demonstrated by this experiment
Name the gas evolved when the following mixtures are heated:
Sodium nitrate and ammonium chloride.
Identify the substance underlined, in the following case:
A solid formed by reaction of two gaes, one of which is acidic and the other basic in nature.
Given one test can be used to detect the presence of the ion produced
What do you observe when Ammonia in excess is mixed with chlorine.
What do you observe when Ammonia solution is added drop by drop and then in excess to aqueous copper sulphate solution.
