Advertisements
Advertisements
प्रश्न
When heated, potassium permanganate decomposes according to the following equation:
\[\ce{2KMnO4 -> \underset{solid residue}{K2MnO4 + MnO2} + O2}\]
Some potassium permanganate was heated in test tube. After collecting one litre of oxygen at room temperature, it was found that the test tube had undergone a loss in mass of 1.32 g. If one litre of hydrogen under the same conditions of temperature and pressure has a mass of 0.0825 g, calculate the relative molecular mass of oxygen.
उत्तर
\[\ce{2KMnO4 -> K2MnO4 + MnO2 + O2}\]
Loss in mass = 1.32 g = 1 lit of oxygen
Vapour density of gas = `"Wt. of certain volume of gas"/"Wt. of same volume of H"_2`
= `1.32/0.0825`
= 16 g
Molecular weight = 2 × Vapour density
= 2 ×16
= 32 g
∴ Relative molecular mass of oxygen is 32 g.
APPEARS IN
संबंधित प्रश्न
Give balanced chemical equations for the following conversions A, B, and C:
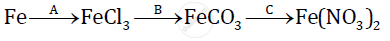
Aluminium carbide reacts with water according to the following equation :
`Al_4C_3 + 12H_2O-> 4Al(OH)_3 + 3CH_4`
1)What mass of aluminium hydroxide is formed from 12g of aluminium carbide?
2) What volume of methane at s.t.p. is obtained from 12g of aluminium carbide?
[Relatively molecular weight of `Al_4Cl_3 = 144; Al(OH)_3 = 78]`
Calculate the relative molecular mass of Ammonium sulphate.
(use K = 39, Cl = 35.5, O = 16, C = 12, H = 1, Na = 23, N = 14, S= 32)
Empirical formula of a compound is CH2O. If its empirical formula is equal to its vapour density, calculate the molecular formula of the compound.
Find the total percentage of oxygen in magnesium nitrate crystal Mg(NO3)2.6H2O.
[H = 1, N = 14, O = 16, Mg = 24]
Calculate the percentage of boron (B) in borax (Na2B4O7.10H2O). [H = 1, B = 11, O = 16, Na = 23],
answer correct to 1 decimal place.
If the relative molecular mass of A is 90, what is the molecular formula of A?
Give two tests of the following:
Oxygen
The atomic mass of Chlorine is 35.5. What is its vapour density?
67.2 litres of hydrogen combines with 44.8 litres of nitrogen to form ammonia under specific conditions as:
\[\ce{N2_{(g)} + 3H2_{(g)} -> 2NH3_{(g)}}\]
Calculate the volume of ammonia produced. What is the other substance, if any, that remains in the resultant mixture?
