Advertisements
Advertisements
प्रश्न
Which among the following statements is incorrect for magnesium metal?
विकल्प
It burns in oxygen with a dazzling white flame
It reacts with cold water to form magnesium oxide and evolves hydrogen gas
It reacts with hot water to form magnesium hydroxide and evolves hydrogen gas
It reacts with steam to form magnesium hydroxide and evolves hydrogen gas
उत्तर
It reacts with cold water to form magnesium oxide and evolves hydrogen gas
Explanation -
Magnesium does not react with cold water. It only reacts with hot water or steam to form magnesium hydroxide and hydrogen gas.
APPEARS IN
संबंधित प्रश्न
Write chemical equation for the event.
Electrolysis of alumina is done.
State what is meant by the ‘reactivity series of metals’
With reference to Water explain with suitable examples of how the reactivity of the metals could be differentiated.
Give a balanced equation for the reversible catalytic reaction involving nitrogen as one of the reactants.
Give a balanced equation for the following type of reaction:
A displacement reaction in which a metal above hydrogen in the reactivity series, displaces another metal from the solution of its compound.
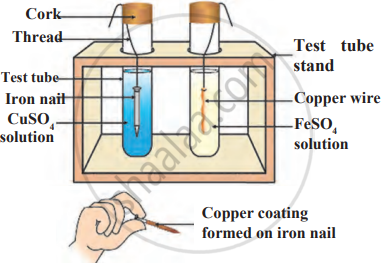
Observe the following diagram and identify the type of reaction and write observation.
Which of the following is the correct arrangement of the given metals in ascending order of their reactivity?
Zinc, Iron, Magnesium, Sodium
Give the steps involved in the extraction of metals of low and medium reactivity from their respective sulphide ores.
Metal ‘A’ has electronic configuration 2, 8, 1 and metal ‘B’ has electronic configuration 2, 8, 8, 2. Out of these, which metal is more reactive? Write the reaction of this metal with dilute HCl acid.
Three metal samples of magnesium, aluminium and iron were taken and rubbed with sandpaper. These samples were then put separately in test tubes containing dilute hydrochloric acid. Thermometers were also suspended in each test tube so that their bulbs dipped in the acid. The rate of formation of bubbles was observed. The above activity was repeated with dilute nitric acid and the observations were recorded.
Answer the following questions:
(i) When the activity was done with dilute hydrochloric acid, then in which one of the test tubes was the rate of formation of bubbles the fastest and the thermometer showed the highest temperature?
(ii) Which metal did not react with dilute hydrochloric acid? Give reason.
(iii) Why is hydrogen gas not evolved when a metal reacts with dilute nitric acid? Name the ultimate products formed in the reaction.
OR
Name the type of reaction on the basis of which the reactivity of metals is decided. You have two metals X and Y. How would you decide which is more reactive than the other?
