Advertisements
Advertisements
प्रश्न
The reaction between acetic acid and sodium carbonate is shown in the following figure.
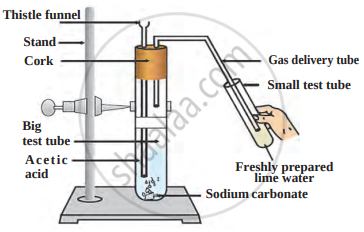
Answer the questions with the help of a diagram.
- Which gas does come out as effervescence in the big test tube?
- What is the colour change in the lime water present in the small test tube?
- Write the related reaction.
उत्तर
- Carbon dioxide gas.
- Lime water turns milky.
- \[\ce{2CH3COOH (aq) + Na2CO3 (g) -> CH3COONa (aq) + H2O (l) + CO2 (g)}\]
APPEARS IN
संबंधित प्रश्न
Ethanoic acid has a .................... odour.
(a) Rotten eggs
(b) Pungent
(c) Vinegar-like
(d) Mild
A student puts a drop of reaction mixture of a saponification reaction first a blue litmus paper and then on a red litmus paper. He may observe that:
(a) There is no change in the blue litmus paper and the red litmus paper turns white.
(b) There is no change in the red litmus paper and the blue litmus paper turns red.
(c) There is no change in the blue litmus paper and the red litmus paper turns blue.
(d) No change in colour is observed in both the litmus papers
A student adds 2 mL of acetic acid to a test tube containing 2 mL of distilled water. He then shakes the test tube well and leaves it to settle for some time. After about 5 minutes he observes that in the test tube there is :
(A) a clear transparent colourless solution
(B) a clear transparent pink solution
(C) a precipitate settling at the bottom of the test tube
(D) a layer of water the layer of acetic acid
A gas is liberated immediately with a brisk effervescence, when you add acetic acid to sodium hydrogen carbonate powder in a test tube. Name the gas and describe the test that confirms the identity of the gas.
Complete the following chemical equations : C2H5OH`("Conc."H_2SO_4)/(443K)`>
When you add a few drops of acetic acid to a test-tube containing sodium bicarbonate powder, which one of the following is your observation?
(A) No reaction takes place
(B) A colourless gas with pungent smell is released with brisk effervescence
(C) A brown coloured gas is released with brisk effervescence
(D) Formation of bubbles of a colourless and odourless gas
Give the common names and IUPAC names of the following compounds of HCOOH.
Write the formulae of methanoic acid.
Ethanol can be oxidized to ethanoic acid. Write the equation and name the oxidizing agent.
Give the structural formulae of acetic acid.
Explain the following term with example.
Monomer
Write the important uses of acetic acid.
State how the following conversions can be carried out:
Ethyl chloride to Ethyl alcohol
State how the following conversions can be carried out:
Ethene to Ethyl alcohol
Choose the correct word/phrase from the options given below to complete the following sentence:
When acetaldehyde is oxidized with acidified potassium dichromate, it forms ______.
When ethanoic acid is treated with NaHCO^ the gas evolved is ______.
In the presence of the acid catalyst, ethanoic acid reacts with ethanol and ______ ester is produced.
A spatula full of sodium carbonate is taken in a test tube and 2 mL of dilute ethanoic acid is added to it.
Suggest a method of testing the gas liberated in the reaction.
