Advertisements
Advertisements
प्रश्न
Which gas is usually liberated when an acid reacts with a metal? Illustrate with an example. How will you test for the presence of this gas?
उत्तर
Hydrogen gas is usually liberated when an acid reacts with a metal.
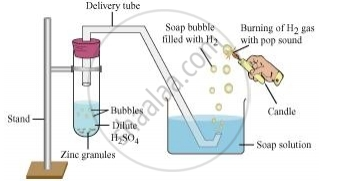
Take few pieces of zinc granules and add 5 ml of dilute H2SO4. Shake it and pass the gas produced into a soap solution. The bubbles of the soap solution are formed. These soap bubbles contain hydrogen gas.
H2SO4+Zn → ZnSO4 + H2 ↑
We can test the evolved hydrogen gas by its burning with a pop sound when a candle is brought near the soap bubbles.
APPEARS IN
संबंधित प्रश्न
Name the gas evolved when dilute HCl reacts with sodium hydrogencarbonate. How is it recognised?
Give the names and formulae of two strong acids and two weak acids.
Give two examples each of organic acids and mineral acids.
One of the following is not an organic acid. This is:
(a) ethanoic acid
(b) formic acid
(c) citric acid
(d) carbonic acid
A white shirt has a yellow stain of curry. When soap is rubbed on this shirt during washing, the yellow stain turns reddish-brown. On rinsing the shirt with plenty of water, the reddish-brown stain turns yellow again.
Name the natural indicator present in curry stain.
What happens during a wasp sting? What is its remedy?
One of the following is a medicine for indigestion. This is:
(a) sodium hydroxide
(b) manganese hydroxide
(c) magnesium hydroxide
(d) potassium hydroxide
Write balanced equation to satisfy the following statement:
\[\ce{Acid + Base -> Salt + water}\]
Out of three metals P, Q and R, P is less reactive than Q and R is more reactive than P and Q both. Suggest an activity to arragne P, Q and R in order of their decreasing reactivity.
What do you understand by the strength of an acid? On what factors does the strength of an acid depend?
Name the positive ion formed When an acid is dissolved in water.
The word sour refers to ______ in Latin.
Most of the acids are not soluble in water.
Two acids ‘A’ and ‘B’ are given. Acid A gives one hydrogen ion per molecule of the acid in solution. Acid B gives two hydrogen ions per molecule of the acid in solution.
- Find out acid A and acid B.
- Which acid is called the King of Chemicals?
Which of the following is acidic in nature?
Name the acid present in the given table.
Which of the following metal does not react with sodium hydroxides?
A metal 'M' on reacting with dilute acid liberates a gas 'G'. The same metal also liberates gas 'G' when reacts with a base.
- Write the name of gas 'G'.
- How will you test the presence of this gas?
- Write chemical equations for the reactions of the metal with (1) an acid and (2) a base.
Name the acid present in grapes.
Name the acid present in lemon.
