Advertisements
Advertisements
Question
Which gas is usually liberated when an acid reacts with a metal? Illustrate with an example. How will you test for the presence of this gas?
Solution
Hydrogen gas is usually liberated when an acid reacts with a metal.
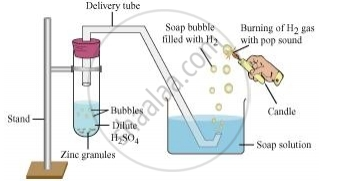
Take few pieces of zinc granules and add 5 ml of dilute H2SO4. Shake it and pass the gas produced into a soap solution. The bubbles of the soap solution are formed. These soap bubbles contain hydrogen gas.
H2SO4+Zn → ZnSO4 + H2 ↑
We can test the evolved hydrogen gas by its burning with a pop sound when a candle is brought near the soap bubbles.
APPEARS IN
RELATED QUESTIONS
Name an indicator which is pink in alkaline solution but turns colourless in acidic solution.
Name the gas evolved when dilute HCl reacts with sodium hydrogencarbonate. How is it recognised?
Why do HCl, H2SO4, HNO3, etc., show acid character in aqueous solutions while solutions of compounds like C6H12O6 (glucose) and C2H5OH (alcohol) do not show acidic character?
One of the following is not an organic acid. This is:
(a) ethanoic acid
(b) formic acid
(c) citric acid
(d) carbonic acid
A white shirt has a yellow stain of curry. When soap is rubbed on this shirt during washing, the yellow stain turns reddish-brown. On rinsing the shirt with plenty of water, the reddish-brown stain turns yellow again.
Name the natural indicator present in curry stain.
One of the following is a medicine for indigestion. This is:
(a) sodium hydroxide
(b) manganese hydroxide
(c) magnesium hydroxide
(d) potassium hydroxide
Bee sting contains:
(a) an acidic liquid
(b) a salt solution
(c) an alkaline liquid
(d) an alcohol
Name a sodium compound which is a constituent of many dry soap powders.
Water is never added to acid in order to dilute it why?
Give one example in the following case:
A hydrogen containing compound which is not an acid.
Tartaric acid is a ______ acid.
Write a balanced reaction for the following conversions (A, B, C, D)
\[\ce{Fe→[A]FeCl2 →[B]FeCO3→[C]Fe(NO3)2→[D]Fe(OH)2}\]
Name the positive ion formed When an acid is dissolved in water.
Acids are bitter in taste.
Phenolphthalein is a synthetic indicator and its colours in acidic and basic solutions, respectively are
You are provided with four test tubes containing sugar solution, baking soda solution, tamarind solution, and salt solution. Write down the activity to find the nature (acidic/basic/neutral) of each solution.
Acids have a sour taste and they are soapy to touch.
Name the acid present in the given table.
Complete the following equation.
ZnO + 2HCl → ______ + ______.
Name the acid present in grapes.
