Advertisements
Advertisements
प्रश्न
Which gas is usually liberated when an acid reacts with a metal? Illustrate with an example. How will you test for the presence of this gas?
उत्तर
Hydrogen gas is usually liberated when an acid reacts with a metal.
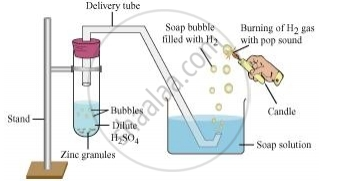
Take few pieces of zinc granules and add 5 ml of dilute H2SO4. Shake it and pass the gas produced into a soap solution. The bubbles of the soap solution are formed. These soap bubbles contain hydrogen gas.
H2SO4+Zn → ZnSO4 + H2 ↑
We can test the evolved hydrogen gas by its burning with a pop sound when a candle is brought near the soap bubbles.
APPEARS IN
संबंधित प्रश्न
When a solution is added to a cloth strip treated with onion extract, then the smell of onion cannot be detected. State whether the given solution contains an acid or a base.\
State some of the uses of mineral acids in industry.
Why is it wrong to treat a wasp sting with baking soda solution?
The acid produced naturally in our stomach is:
(a) acetic acid
(b) citric acid
(c) hydrochloric acid
(d) sulphuric acid
Name a sodium compound which is a constituent of many dry soap powders.
Out of three metals P, Q and R, P is less reactive than Q and R is more reactive than P and Q both. Suggest an activity to arragne P, Q and R in order of their decreasing reactivity.
Tartaric acid is a ______ acid.
Complete the following table and write one equation for each to justify the statement:
|
Reactants |
Products |
Method |
|
Soluble base + Acid (dil) |
Salt + water |
Neutralisation Titration |
|
Metal + Non-metal |
Salt (soluble/insoluble) |
______ |
|
Insoluble base + ______ |
Salt (soluble) + water |
______ |
|
Active metal + Acid (dil) |
______ + ______ |
______ |
|
Soluble salt solution (A) + Soluble salt solution (B) |
Precipitated salt + Soluble salt |
______ |
|
Carbonate/bicarbonate + Acid (dil) |
Salt + ______ + ______ |
Decomposition of carbonate |
|
Chlorides/nitrates + Acid (conc.) |
______ + ______ |
Decomposition of chlorides and nitrates |
Write the balanced equation for the preparation of the following compound (as major product) starting from iron and using only one other substance:
Iron (III) chloride
The word sour refers to ______ in Latin.
Acids are corrosive in nature.
Heshna and Keerthi are friends. Keerthi’s teeth are white without caries, but Heshna has teeth with caries. Why? How is it formed?
Zn + 2 HCl → ZnCl2 +_____↑
Match the acids given in column (A) with their correct source given in column (B)
| Column (A) | Column (B) |
| (a) Lactic acid | Tamarind |
| (b) Acetic acid | Lemon |
| (c) Citric acid | Vinegar |
| (d) Tartaric acid | Curd |
The correct way of making a solution of acid in water is to
Phenolphthalein is a synthetic indicator and its colours in acidic and basic solutions, respectively are
A farmer was unhappy because of his low crop yield. He discussed the problem with an agricultural scientist and realised that the soil of his field was either too acidic or too basic. What remedy would you suggest to the farmer to neutralise the soil?
Write the important uses of hydrochloric acid and sulphuric acids.
Which of the following metal does not react with sodium hydroxides?
