Advertisements
Advertisements
प्रश्न
Why should curd and sour substances not be kept in brass and copper vessels?
उत्तर १
Curd and other sour substances contain acids. Therefore, when they are kept in brass and copper vessels, the metal reacts with the acid to liberate hydrogen gas and harmful products, thereby spoiling the food.
उत्तर २
Curd and sour food substances contain acids; these acidic substances have a possibility of reacting with the metal. If this reaction takes place then it can cause food poisoning and damage people’s health.
APPEARS IN
संबंधित प्रश्न
Why do HCl, HNO3, etc., show acidic characters in aqueous solutions while solutions of compounds like alcohol and glucose do not show acidic character?
How is the concentration of hydroxide ions (OH−) affected when excess base is dissolved in a solution of sodium hydroxide?
Equal lengths of magnesium ribbons are taken in test tubes A and B. Hydrochloric acid (HCl) is added to test tube A, while acetic acid (CH3COOH) is added to test tube B. In which test tube will the fizzing occur more vigorously, and why?
Complete and balance the following chemical equations
Na2 CO3 (s) + HCI (aq) →
Complete and balance the followingchemicalequations:
NaHCO3 (S) + HCI (aq) →
Complete and balance the following chemical equations:
NaOH (aq) HCI (aq)→
Give chemical equation of the reaction involved.
A solution reacts with zinc granules to give a gas which burns with a 'pop' sound. The solution contains:
(a) Mg(OH)2
(b) Na2CO3
(c) NaCl
(d) HCl
On adding dilute hydrochloric acid to copper oxide powder, the solution formed is blue-green.
Predict the new compound formed which imparts a blue-green colour to solution.
A substance X which is used as an antacid reacts with dilute hydrochloric acid to produce a gas Y which is used in one type of fire-extinguisher. Name the substance X and gas Y. Write a balanced equation for the chemical reaction which takes place.
Write the chemical formula of sodium carbonate decahydrate.
State one use of bleaching powder (other than bleaching).
Name a metal compound which has detergent properties (cleansing properties).
Write the chemical formula of washing soda. How can it be obtained from baking soda? List two industries in which washing soda is used for other purposes than washing clothes.
Lime water reacts with chlorine to form:
The table given below shows the reaction of a few elements with acids and bases to evolve Hydrogen gas.
| Element | Acid | Base |
| A |
✗
|
✗ |
| B |
✓
|
✓ |
| C | ✓ | ✗ |
| D | ✓ | ✓ |
Which of these elements form amphoteric oxides?
In an attempt to demonstrate electrical conductivity through an electrolyte, the apparatus setup. Which among the following statement(s) is(are) correct?
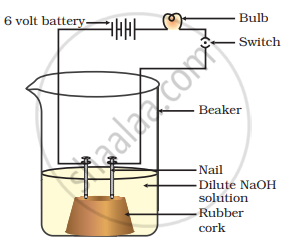
- Bulb will not glow because electrolyte is not acidic
- Bulb will glow because NaOH is a strong base and furnishes ions for conduction.
- Bulb will not glow because circuit is incomplete
- Bulb will not glow because it depends upon the type of electrolytic solution
In the following schematic diagram for the preparation of hydrogen gas as shown in the figure, what would happen if following changes are made?

- In place of zinc granules, same amount of zinc dust is taken in the test tube
- Instead of dilute sulphuric acid, dilute hydrochloric acid is taken
- In place of zinc, copper turnings are taken
- Sodium hydroxide is taken in place of dilute sulphuric acid and the tube is heated.
A substance 'X' is used as a building material and is insoluble in water. When it reacts with dil. HCl, it produces a gas which turns lime water milky.
- Write the chemical name and formula of 'X'.
- Write chemical equations for the chemical reactions involved in the above statements.
