Advertisements
Advertisements
प्रश्न
Which metal is extracted from cinnabar ore?
उत्तर
Mercury is extracted from cinnabar ore. It is the sulphide ore of mercury and its chemical formula is HgS. The name of the compound present in cinnabar is mercury (II) sulphide. It is a sulphide ore; therefore, it is first heated in the presence of surplus air (roasting). This converts mercury (II) sulphide to mercury (II) oxide.

Mercury (II) oxide is then heated again to around 300°C On heating, it gets reduced to mercury.
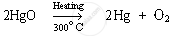
APPEARS IN
संबंधित प्रश्न
In the extraction of aluminium: Name the process of concentration of bauxite.
In the extraction of aluminium Write the cathode reaction in electrolytic reduction of alumina.
Manganese metal is extracted from manganese dioxide by a reduction process by making use of:
(a) carbon
(b) hydrogen
(c) electrolysis
(d) aluminium
Give reasons, why aluminum is used in:
In aluminiothermy
State one observation for each of the following :
Copper sulphate solution is electrolysed using copper electrodes.
Complete the incomplete statement with missing word:
Metals form positive ions while non- metals ______.
In the electrolytic reduction of alumina, the lining of graphite acts as an anode.
Explain the following reaction with the balanced equation.
Zinc carbonate is heated strongly in limited supply of air
2 mL each of concentrated HCl, HNO3 and a mixture of concentrated HCl and concentrated HNO3 in the ratio of 3 : 1 were taken in test tubes labelled as A, B and C. A small piece of metal was put in each test tube. No change occurred in test tubes A and B but the metal got dissolved in test tube C respectively. The metal could be
Iqbal treated a lustrous, divalent element M with sodium hydroxide. He observed the formation of bubbles in reaction mixture. He made the same observations when this element was treated with hydrochloric acid. Suggest how can he identify the produced gas. Write chemical equations for both the reactions.
