Advertisements
Advertisements
प्रश्न
Which metal is extracted from cinnabar ore?
उत्तर
Mercury is extracted from cinnabar ore. It is the sulphide ore of mercury and its chemical formula is HgS. The name of the compound present in cinnabar is mercury (II) sulphide. It is a sulphide ore; therefore, it is first heated in the presence of surplus air (roasting). This converts mercury (II) sulphide to mercury (II) oxide.

Mercury (II) oxide is then heated again to around 300°C On heating, it gets reduced to mercury.
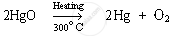
APPEARS IN
संबंधित प्रश्न
M is a metal above hydrogen in the activity series and its oxide has the formula M2O. This oxide when dissolved in water forms the corresponding hydroxide which is a good conductor of electricity. In the above context, answer the following:
1) What kind of combination exists between M and O?
2)How many electrons are there in the outermost shell of M?
3) Name the group to which M belongs.
4) State the reaction taking place at the cathode.
5) Name the product at the anode.
Explain why, carbon cannot reduce oxides of sodium or magnesium.
Define the term mineral.
Which metal can be extracted from the following ore?
Haematite
Give the principles of electromagnetic separation.
Name the ore of mercury. With the help of balanced chemical equations, explain the process of extraction of mercury from its ore.
Write the name.
Metals which are amphoteric in nature.
Explain concept with example/explain with the help of a balanced equation.
Ionic bond and ionic compounds
Explain concept with example/explain with the help of a balanced equation.
Calcination
The given reaction shows one of the processes to extract the metals like Iron and Manganese.
\[\ce{MnO2(s) + Al(s) -> Mn(l) + Al2O3(s) + Heat}\]
Give a reason why Aluminium is preferably used in thermite reactions.
