Advertisements
Advertisements
Question
Which metal is extracted from cinnabar ore?
Solution
Mercury is extracted from cinnabar ore. It is the sulphide ore of mercury and its chemical formula is HgS. The name of the compound present in cinnabar is mercury (II) sulphide. It is a sulphide ore; therefore, it is first heated in the presence of surplus air (roasting). This converts mercury (II) sulphide to mercury (II) oxide.

Mercury (II) oxide is then heated again to around 300°C On heating, it gets reduced to mercury.
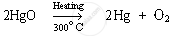
APPEARS IN
RELATED QUESTIONS
Metallic oxides of zinc, magnesium and copper were heated with the following metals.
| Metal | Zinc | Magnesium | Copper |
| Zinc oxide | – | – | – |
| Magnesium oxide | – | – | – |
| Copper oxide | – | – | – |
In which cases will you find displacement reactions taking place?
Give reasons.
Aluminium is a highly reactive metal, yet it is used to make utensils for cooking.
How is aluminium metal extracted? Explain with the help of an equation.
Which metal is extracted from calamine ore?
Which of the following metals are extracted by the electrolysis of their molten chlorides?
(a) Na and Hg
(b) Hg and Mg
(c) Na and Mg
(d) Cu and Fe
Give reasons, why aluminum is used in:
In making ships
Explain the following reaction with the balanced equation.
Chlorine dissolved in water
Explain the following reaction with the balanced equation.
Zinc carbonate is heated strongly in limited supply of air
Why should the metal sulphides and carbonates be converted to metal oxides in the process of extraction of metal from them?
On the basis of reactivity metals are grouped into three categories:
- Metals of low reactivity
- Metals of medium reactivity
- Metals of high reactivity
Therefore metals are extracted in pure form from their ores on the basis of their chemical properties.
Metals of high reactivity are extracted from their ores by electrolysis of the molten ore.
Metals of low reactivity are extracted from their sulphide ores, which are converted into their oxides. The oxides of these metals are reduced to metals by simple heating.
(a) Name the process of reduction used for a metal that gives vigorous reaction with air and water both.
(b) Carbon cannot be used as a reducing agent to obtain aluminium from its oxide? Why?
(c) Describe briefly the method to obtain mercury from cinnabar. Write the chemical equation for the reactions involved in the process.
OR
(c) Differentiate between roasting and calcination giving chemical equation for each.
