Advertisements
Advertisements
Question
How is aluminium metal extracted? Explain with the help of an equation.
Solution
Aluminium is extracted by electrolytic reduction of its molten oxide. Aluminium is a highly reactive metal and it is placed at the top of the reactivity series. This cannot be reduced by other reducing agents such as coke, carbon monoxide etc.
On passing electricity through molten aluminium oxide, decomposition reaction occurs and formation of aluminium metal and oxygen gas takes place.

Molten aluminium oxide consists of aluminium and oxide ions. The reactions that occur during electrolysis are:
1. Cathode produces electrons to reduce aluminium ions to aluminium atoms (or aluminium metal) by acting as reducing agent. Aluminium ions are cations; therefore, they get attracted to negatively charged cathode and get deposited there.
Cathode: 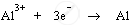
xide ions are anions; therefore, they get attracted to positively charged anode. These oxide ions are oxidised to oxygen gas. Oxygen gas is produced at anode.
Anode: 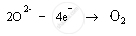
APPEARS IN
RELATED QUESTIONS
Name one metal which is extracted by reduction with carbon.
Name one ore of aluminium. Name the aluminium compound present in this ore and write its chemical formula.
The articles made of silver metal become dark on prolonged exposure to air. This is due to the formation of a layer of its:
(a) oxide
(b) hydride
(c) sulphide
(d) carbonate
Give the chemical formula of :
Bauxite
Give the chemical formula of :
Cryolite
Which method of refining is used to get 99.8% pure aluminium?
State three objectives achieved during the roasting of ores.
How is the method of extraction of metals high up in the reactivity series different from that for metals in the middle? Why can the same process not be applied for them? Name the process used for the extraction of these metals.
Bauxite reacts with sodium hydroxide in the Bayer’s process.
Explain Bayer’s process of concentration of bauxite with a chemical equation.
