Advertisements
Advertisements
प्रश्न
Which of the following defects decrease the density?
(i) Interstitial defect
(ii) Vacancy defect
(iii) Frankel defect
(iv) Schottky defect
उत्तर
(ii) Vacancy defect
(iv) Schottky defect
Explanation:
Vacancy and Schottky defect which lead to decrease the density both are the types of a stoichiometric defect. In case of Frenkel defect and interstitial defect, there is no change in density of substance.
APPEARS IN
संबंधित प्रश्न
Explain the following terms with suitable examples : Interstitials
Ionic solids containing large differences in sizes of ions show ____________.
Defect shown by non-ionic solids is ____________.
The following diagram shows:
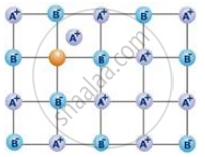
Schottky defect defines imperfection in the lattice structure of ____________.
Which is not correct about the Schottky defects?
Schottky defect is observed in crystals when ______.
Which of the following point defects are shown by AgBr(s) crystals?
- Schottky defect
- Frenkel defect
- Metal excess defect
- Metal deficiency defect
Which of the following defects is also known as dislocation defect?
Match the defects given in Column I with the statements in given Column II.
| Column I | Column II |
| (i) Simple vacancy defect | (a) shown by non-ionic solids and increases density of the solid. |
| (ii) Simple interstitial defect | (b) shown by ionic solids and decreases density of the solid. |
| (iii) Frenkel defect | (c) shown by non ionic solids and density of the solid decreases. |
| (iv) Schottky defect | (d) shown by ionic solids and density of the solid remains the same. |
